Radioiodinated Capsids Facilitate In Vivo Non-Invasive Tracking of Adeno-Associated Gene Transfer Vectors
- PMID: 28059103
- PMCID: PMC5216390
- DOI: 10.1038/srep39594
Radioiodinated Capsids Facilitate In Vivo Non-Invasive Tracking of Adeno-Associated Gene Transfer Vectors
Abstract
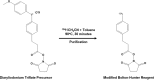
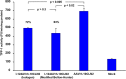
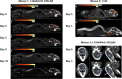
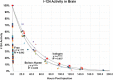
Viral vector mediated gene therapy has become commonplace in clinical trials for a wide range of inherited disorders. Successful gene transfer depends on a number of factors, of which tissue tropism is among the most important. To date, definitive mapping of the spatial and temporal distribution of viral vectors in vivo has generally required postmortem examination of tissue. Here we present two methods for radiolabeling adeno-associated virus (AAV), one of the most commonly used viral vectors for gene therapy trials, and demonstrate their potential usefulness in the development of surrogate markers for vector delivery during the first week after administration. Specifically, we labeled adeno-associated virus serotype 10 expressing the coding sequences for the CLN2 gene implicated in late infantile neuronal ceroid lipofuscinosis with iodine-124. Using direct (Iodogen) and indirect (modified Bolton-Hunter) methods, we observed the vector in the murine brain for up to one week using positron emission tomography. Capsid radioiodination of viral vectors enables non-invasive, whole body, in vivo evaluation of spatial and temporal vector distribution that should inform methods for efficacious gene therapy over a broad range of applications.
Figures


Similar articles
-
Clinical protocol. Administration of a replication-deficient adeno-associated virus gene transfer vector expressing the human CLN2 cDNA to the brain of children with late infantile neuronal ceroid lipofuscinosis.Hum Gene Ther. 2004 Nov;15(11):1131-54. doi: 10.1089/hum.2004.15.1131. Hum Gene Ther. 2004. PMID: 15610613 Clinical Trial.
-
Long-term expression and safety of administration of AAVrh.10hCLN2 to the brain of rats and nonhuman primates for the treatment of late infantile neuronal ceroid lipofuscinosis.Hum Gene Ther Methods. 2012 Oct;23(5):324-35. doi: 10.1089/hgtb.2012.120. Epub 2012 Nov 6. Hum Gene Ther Methods. 2012. PMID: 23131032 Free PMC article.
-
An AAV variant selected through NHP screens robustly transduces the brain and drives secreted protein expression in NHPs and mice.Sci Transl Med. 2025 May 14;17(798):eadr2531. doi: 10.1126/scitranslmed.adr2531. Epub 2025 May 14. Sci Transl Med. 2025. PMID: 40367194
-
CLN2 Disease (Classic Late Infantile Neuronal Ceroid Lipofuscinosis).Pediatr Endocrinol Rev. 2016 Jun;13 Suppl 1:682-8. Pediatr Endocrinol Rev. 2016. PMID: 27491216 Review.
-
The role of the adeno-associated virus capsid in gene transfer.Methods Mol Biol. 2008;437:51-91. doi: 10.1007/978-1-59745-210-6_2. Methods Mol Biol. 2008. PMID: 18369962 Free PMC article. Review.
Cited by
-
A novel approach to quantitate biodistribution and transduction of adeno-associated virus gene therapy using radiolabeled AAV vectors in mice.Mol Ther Methods Clin Dev. 2024 Aug 19;32(3):101326. doi: 10.1016/j.omtm.2024.101326. eCollection 2024 Sep 12. Mol Ther Methods Clin Dev. 2024. PMID: 39286334 Free PMC article.
-
Positron emission tomography imaging of novel AAV capsids maps rapid brain accumulation.Nat Commun. 2020 Apr 30;11(1):2102. doi: 10.1038/s41467-020-15818-4. Nat Commun. 2020. PMID: 32355221 Free PMC article.
-
Radioanalytical Techniques to Quantitatively Assess the Biological Uptake and In Vivo Behavior of Hazardous Substances.Molecules. 2020 Sep 1;25(17):3985. doi: 10.3390/molecules25173985. Molecules. 2020. PMID: 32882977 Free PMC article. Review.
-
Recent developments in translational imaging of in vivo gene therapy outcomes.Mol Ther. 2025 Jun 4;33(6):2548-2564. doi: 10.1016/j.ymthe.2024.12.049. Epub 2024 Dec 30. Mol Ther. 2025. PMID: 39741403 Review.
-
Radiolabeling Strategies of Nanobodies for Imaging Applications.Diagnostics (Basel). 2021 Aug 25;11(9):1530. doi: 10.3390/diagnostics11091530. Diagnostics (Basel). 2021. PMID: 34573872 Free PMC article. Review.
References
-
- Journal of Gene Medicine, Gene Therapy Clinical Trials Worldwide, http://www.abedia.com/wiley/vectors.php Date of access: 11/08/2016 (2016).
-
- Wilhem-Merten P., & Al-Rubeai M., Eds Viral Vectors for Gene Therapy: Methods and Protocols. (Humana Press, Springer Science, 2011).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

