Using imaging as a biomarker for asthma
- PMID: 28065276
- PMCID: PMC5224930
- DOI: 10.1016/j.jaci.2016.11.009
Using imaging as a biomarker for asthma
Abstract
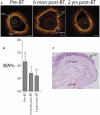
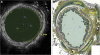
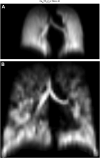
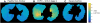
There have been significant advancements in the various imaging techniques being used for the evaluation of asthmatic patients, both from a clinical and research perspective. Imaging characteristics can be used to identify specific asthmatic phenotypes and provide a more detailed understanding of endotypes contributing to the pathophysiology of the disease. Computed tomography, magnetic resonance imaging, and positron emission tomography can be used to assess pulmonary structure and function. It has been shown that specific airway and lung density measurements using computed tomography correlate with clinical parameters, including severity of disease and pathology, but also provide unique phenotypes. Hyperpolarized 129Xe and 3He are gases used as contrast media for magnetic resonance imaging that provide measurement of distal lung ventilation reflecting small-airway disease. Positron emission tomography can be useful to identify and target lung inflammation in asthmatic patients. Furthermore, imaging techniques can serve as a potential biomarker and be used to assess response to therapies, including newer biological treatments and bronchial thermoplasty.
Keywords: Imaging; MRI; PET; biomarker; chest CT.
Copyright © 2016 American Academy of Allergy, Asthma & Immunology. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Hartley R, Baldi S, Brightling C, Gupta S. Novel imaging approaches in adult asthma and their clinical potential. Expert Rev. Clin. Immunol. 2015;11(10):1147–1162. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

