Proteolytic properties of single-chain factor XII: a mechanism for triggering contact activation
- PMID: 28069606
- PMCID: PMC5356457
- DOI: 10.1182/blood-2016-10-744110
Proteolytic properties of single-chain factor XII: a mechanism for triggering contact activation
Abstract
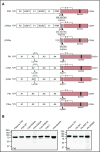
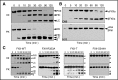
When blood is exposed to variety of artificial surfaces and biologic substances, the plasma proteins factor XII (FXII) and prekallikrein undergo reciprocal proteolytic conversion to the proteases αFXIIa and α-kallikrein by a process called contact activation. These enzymes contribute to host-defense responses including coagulation, inflammation, and fibrinolysis. The initiating event in contact activation is debated. To test the hypothesis that single-chain FXII expresses activity that could initiate contact activation, we prepared human FXII variants lacking the Arg353 cleavage site required for conversion to αFXIIa (FXII-R353A), or lacking the 3 known cleavage sites at Arg334, Arg343, and Arg353 (FXII-T, for "triple" mutant), and compared their properties to wild-type αFXIIa. In the absence of a surface, FXII-R353A and FXII-T activate prekallikrein and cleave the tripeptide S-2302, demonstrating proteolytic activity. The activity is several orders of magnitude weaker than that of αFXIIa. Polyphosphate, an inducer of contact activation, enhances PK activation by FXII-T, and facilitates FXII-T activation of FXII and FXI. In plasma, FXII-T and FXII-R353A, but not FXII lacking the active site serine residue (FXII-S544A), shortened the clotting time of FXII-deficient plasma and enhanced thrombin generation in a surface-dependent manner. The effect was not as strong as for wild-type FXII. Our results support a model for induction of contact activation in which activity intrinsic to single-chain FXII initiates αFXIIa and α-kallikrein formation on a surface. αFXIIa, with support from α-kallikrein, subsequently accelerates contact activation and is responsible for the full procoagulant activity of FXII.
© 2017 by The American Society of Hematology.
Figures






Comment in
-
Contact ignition by single-chain XIIa.Blood. 2017 Mar 16;129(11):1411-1412. doi: 10.1182/blood-2017-02-763532. Blood. 2017. PMID: 28302690 No abstract available.
References
-
- Macfarlane RG. An enzyme cascade in the blood clotting mechanism, and its function as a biochemical amplifier. Nature. 1964;202:498-499. - PubMed
-
- Davie EW, Ratnoff OD. Waterfall sequence for intrinsic blood clotting. Science. 1964;145(3638):1310-1312. - PubMed
-
- Long AT, Kenne E, Jung R, Fuchs TA, Renné T. Contact system revisited: an interface between inflammation, coagulation, and innate immunity. J Thromb Haemost. 2016;14(3):427-437. - PubMed
-
- Schmaier AH. The contact activation and kallikrein/kinin systems: pathophysiologic and physiologic activities. J Thromb Haemost. 2016;14(1):28-39. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

