Myosin phosphatase and RhoA-activated kinase modulate arginine methylation by the regulation of protein arginine methyltransferase 5 in hepatocellular carcinoma cells
- PMID: 28074910
- PMCID: PMC5225440
- DOI: 10.1038/srep40590
Myosin phosphatase and RhoA-activated kinase modulate arginine methylation by the regulation of protein arginine methyltransferase 5 in hepatocellular carcinoma cells
Abstract
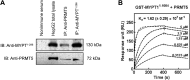
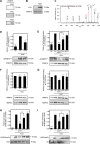
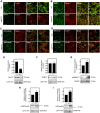
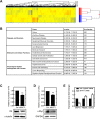
Myosin phosphatase (MP) holoenzyme is a protein phosphatase-1 (PP1) type Ser/Thr specific enzyme that consists of a PP1 catalytic (PP1c) and a myosin phosphatase target subunit-1 (MYPT1). MYPT1 is an ubiquitously expressed isoform and it targets PP1c to its substrates. We identified the protein arginine methyltransferase 5 (PRMT5) enzyme of the methylosome complex as a MYPT1-binding protein uncovering the nuclear MYPT1-interactome of hepatocellular carcinoma cells. It is shown that PRMT5 is regulated by phosphorylation at Thr80 by RhoA-associated protein kinase and MP. Silencing of MYPT1 increased the level of the PRMT5-specific symmetric dimethylation on arginine residues of histone 2 A/4, a repressing gene expression mark, and it resulted in a global change in the expression of genes affecting cellular processes like growth, proliferation and cell death, also affecting the expression of the retinoblastoma protein and c-Myc. The phosphorylation of the MP inhibitory MYPT1T850 and the regulatory PRMT5T80 residues as well as the symmetric dimethylation of H2A/4 were elevated in human hepatocellular carcinoma and in other types of cancers. These changes correlated positively with the grade and state of the tumors. Our results suggest the tumor suppressor role of MP via inhibition of PRMT5 thereby regulating gene expression through histone arginine dimethylation.
Figures

References
-
- Nagao M. et al. Role of protein phosphatases in malignant transformation. Princess Takamatsu symposia 20, 177–184 (1989). - PubMed
-
- Dombradi V., Axton J. M., Barker H. M. & Cohen P. T. Protein phosphatase 1 activity in Drosophila mutants with abnormalities in mitosis and chromosome condensation. FEBS Lett 275, 39–43 (1990). - PubMed
-
- Berndt N. Protein dephosphorylation and the intracellular control of the cell number. Frontiers in bioscience: a journal and virtual library 4, D22–42 (1999). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

