Adenosine receptors regulate gap junction coupling of the human cerebral microvascular endothelial cells hCMEC/D3 by Ca2+ influx through cyclic nucleotide-gated channels
- PMID: 28075020
- PMCID: PMC5390872
- DOI: 10.1113/JP273150
Adenosine receptors regulate gap junction coupling of the human cerebral microvascular endothelial cells hCMEC/D3 by Ca2+ influx through cyclic nucleotide-gated channels
Abstract
Key points: Gap junction channels are essential for the formation and regulation of physiological units in tissues by allowing the lateral cell-to-cell diffusion of ions, metabolites and second messengers. Stimulation of the adenosine receptor subtype A2B increases the gap junction coupling in the human blood-brain barrier endothelial cell line hCMEC/D3. Although the increased gap junction coupling is cAMP-dependent, neither the protein kinase A nor the exchange protein directly activated by cAMP were involved in this increase. We found that cAMP activates cyclic nucleotide-gated (CNG) channels and thereby induces a Ca2+ influx, which leads to the increase in gap junction coupling. The report identifies CNG channels as a possible physiological link between adenosine receptors and the regulation of gap junction channels in endothelial cells of the blood-brain barrier.
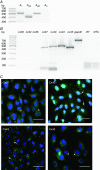
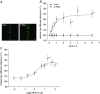
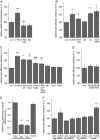
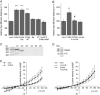
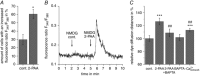
Abstract: The human cerebral microvascular endothelial cell line hCMEC/D3 was used to characterize the physiological link between adenosine receptors and the gap junction coupling in endothelial cells of the blood-brain barrier. Expressed adenosine receptor subtypes and connexin (Cx) isoforms were identified by RT-PCR. Scrape loading/dye transfer was used to evaluate the impact of the A2A and A2B adenosine receptor subtype agonist 2-phenylaminoadenosine (2-PAA) on the gap junction coupling. We found that 2-PAA stimulated cAMP synthesis and enhanced gap junction coupling in a concentration-dependent manner. This enhancement was accompanied by an increase in gap junction plaques formed by Cx43. Inhibition of protein kinase A did not affect the 2-PAA-related enhancement of gap junction coupling. In contrast, the cyclic nucleotide-gated (CNG) channel inhibitor l-cis-diltiazem, as well as the chelation of intracellular Ca2+ with BAPTA, or the absence of external Ca2+ , suppressed the 2-PAA-related enhancement of gap junction coupling. Moreover, we observed a 2-PAA-dependent activation of CNG channels by a combination of electrophysiology and pharmacology. In conclusion, the stimulation of adenosine receptors in hCMEC/D3 cells induces a Ca2+ influx by opening CNG channels in a cAMP-dependent manner. Ca2+ in turn induces the formation of new gap junction plaques and a consecutive sustained enhancement of gap junction coupling. The report identifies CNG channels as a physiological link that integrates gap junction coupling into the adenosine receptor-dependent signalling of endothelial cells of the blood-brain barrier.
Keywords: CNG channel; Ca2+ influx; adenosine receptor; blood-brain barrier; cyclic adenosine monophosphate (cAMP); endothelial cell; gap junction.
© 2017 The Authors. The Journal of Physiology © 2017 The Physiological Society.
Figures

Comment in
-
Unravelling a novel mechanism for the up-regulation of connexin43 gap junctions between cells derived from the blood-brain barrier.J Physiol. 2017 Apr 15;595(8):2411-2412. doi: 10.1113/JP274027. Epub 2017 Mar 20. J Physiol. 2017. PMID: 28205211 Free PMC article. No abstract available.
References
-
- Atkinson MM, Lampe PD, Lin HH, Kollander R, Li XR & Kiang DT (1995). Cyclic AMP modifies the cellular distribution of connexin43 and induces a persistent increase in the junctional permeability of mouse mammary tumor cells. J Cell Sci 108, 3079–3090. - PubMed
-
- Begandt D, Bader A, Gerhard L, Lindner J, Dreyer L, Schlingmann B & Ngezahayo A (2013. b). Dipyridamole‐related enhancement of gap junction coupling in the GM‐7373 aortic endothelial cells correlates with an increase in the amount of connexin 43 mRNA and protein as well as gap junction plaques. J Bioenerg Biomembr 45, 409–419. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

