The potential of spectral domain optical coherence tomography imaging based retinal biomarkers
- PMID: 28078103
- PMCID: PMC5220620
- DOI: 10.1186/s40942-016-0054-7
The potential of spectral domain optical coherence tomography imaging based retinal biomarkers
Abstract
Background: Biomarker", a merged word of "biological marker", refers to a broad subcategory of medical signs that objectively indicate the state of health, and well-being of an individual. Biomarkers hold great promise for personalized medicine as information gained from diagnostic or progression markers can be used to tailor treatment to the individual for highly effective intervention in the disease process. Optical coherence tomography (OCT) has proved useful in identifying various biomarkers in ocular and systemic diseases.
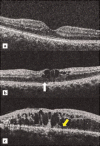
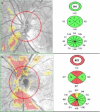
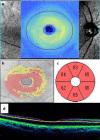
Main body: Spectral domain optical coherence tomography imaging-based biomarkers provide a valuable tool for detecting the earlier stages of the disease, tracking progression, and monitoring treatment response. The aim of this review article is to analyze various OCT based imaging biomarkers and their potential to be considered as surrogate endpoints for diabetic retinopathy, age related macular degeneration, retinitis pigmentosa and vitreomacular interface disorder. These OCT based surrogate markers have been classified as retinal structural alterations (macular central subfield thickness and cube average thickness); retinal ultrastructural alterations (disruption of external limiting membrane and ellipsoid zone, thinning of retinal nerve fiber layer and ganglion cell layer); intraretinal microangiopathic changes; choroidal surrogate endpoints; and vitreoretinal interface endpoints.
Conclusion: OCT technology is changing very quickly and throughout this review there are some of the multiple possibilities that OCT based imaging biomarkers will be more useful in the near future for diagnosis, prognosticating disease progression and as endpoint in clinical trials.
Keywords: Age related macular degeneration; Biomarkers; Diabetic retinopathy; Inherited macular disorder; Optical coherence tomography; Retinitis pigmentosa; Vitreomacular interface disorders.
Figures


Similar articles
-
OCT Biomarkers in Neovascular Age-Related Macular Degeneration: A Narrative Review.J Ophthalmol. 2021 Jul 17;2021:9994098. doi: 10.1155/2021/9994098. eCollection 2021. J Ophthalmol. 2021. PMID: 34336265 Free PMC article. Review.
-
Swept-source optical coherence tomography changes and visual acuity among Palestinian retinitis Pigmentosa patients: a cross-sectional study.BMC Ophthalmol. 2021 Jul 29;21(1):289. doi: 10.1186/s12886-021-02047-6. BMC Ophthalmol. 2021. PMID: 34320936 Free PMC article.
-
Optical coherence tomography in imaging of macular diseases.Klin Oczna. 2010;112(4-6):138-46. Klin Oczna. 2010. PMID: 20825070 Review.
-
Disorganization of retinal inner layers correlates with ellipsoid zone disruption and retinal nerve fiber layer thinning in diabetic retinopathy.J Diabetes Complications. 2019 Aug;33(8):550-553. doi: 10.1016/j.jdiacomp.2019.05.006. Epub 2019 May 17. J Diabetes Complications. 2019. PMID: 31186163
-
Predicting visual outcomes for macular disease using optical coherence tomography.Saudi J Ophthalmol. 2011 Apr;25(2):145-58. doi: 10.1016/j.sjopt.2011.01.003. Epub 2011 Jan 26. Saudi J Ophthalmol. 2011. PMID: 23960916 Free PMC article.
Cited by
-
Retinal relaxation following membrane peeling: Effect on vision, central macular thickness, and vector analysis of motion.J Clin Transl Res. 2020 May 12;5(5):236-242. eCollection 2020 Jun 9. J Clin Transl Res. 2020. PMID: 33029566 Free PMC article.
-
A multidisciplinary approach to inherited retinal dystrophies from diagnosis to initial care: a narrative review with inputs from clinical practice.Orphanet J Rare Dis. 2023 Jul 31;18(1):223. doi: 10.1186/s13023-023-02798-z. Orphanet J Rare Dis. 2023. PMID: 37525225 Free PMC article. Review.
-
Deep learning in optical coherence tomography: Where are the gaps?Clin Exp Ophthalmol. 2023 Nov;51(8):853-863. doi: 10.1111/ceo.14258. Epub 2023 May 28. Clin Exp Ophthalmol. 2023. PMID: 37245525 Free PMC article. Review.
-
[Biomarkers in full-thickness and lamellar defects of the macula].Ophthalmologe. 2021 Apr;118(4):321-336. doi: 10.1007/s00347-021-01340-5. Epub 2021 Mar 1. Ophthalmologe. 2021. PMID: 33646383 Review. German.
-
Retinitis Pigmentosa: Current Clinical Management and Emerging Therapies.Int J Mol Sci. 2023 Apr 19;24(8):7481. doi: 10.3390/ijms24087481. Int J Mol Sci. 2023. PMID: 37108642 Free PMC article. Review.
References
-
- Downing GJ. Biomarkers and surrogate endpoints: clinical research and applications: proceedings of the NIH-FDA conference held on 15–16 April 1999 in Bethesda, Maryland, USA. Elsevier Health Sciences; 2000.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

