Getting to and through the inner nuclear membrane during herpesvirus nuclear egress
- PMID: 28086162
- PMCID: PMC5503802
- DOI: 10.1016/j.ceb.2016.12.007
Getting to and through the inner nuclear membrane during herpesvirus nuclear egress
Abstract
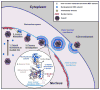
Herpesviruses, like most DNA viruses, replicate and package their genomes into capsids in the host cell nucleus. Capsids then transit to the cytoplasm in a fascinating process called nuclear egress, which includes several unusual steps: Movement of capsids from the nuclear interior to the periphery, disruption of the nuclear lamina, capsid budding through the inner nuclear membrane, and fusion of enveloped particles with the outer nuclear membrane. Here, we review recent advances and emerging questions relating to herpesvirus nuclear egress, emphasizing controversies regarding mechanisms for capsid trafficking to the nuclear periphery, and implications of recent structures of the two-subunit, viral nuclear egress complex for the process, particularly at the step of budding through the inner nuclear membrane.
Copyright © 2016 Elsevier Ltd. All rights reserved.
Figures


References
-
- Pellett PE, Roizman B. Herpesviridae. In: Knipe DM, Howley PM, editors. Fields Virology. 2013. pp. 1802–1822.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

