Mas-related G protein coupled receptor-X2: A potential new target for modulating mast cell-mediated allergic and inflammatory diseases
- PMID: 28090599
- PMCID: PMC5233413
Mas-related G protein coupled receptor-X2: A potential new target for modulating mast cell-mediated allergic and inflammatory diseases
Abstract
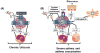
Mast cells (MCs) are tissue resident immune cells that are best known for their roles in allergic and inflammatory diseases. In addition to the high affinity IgE receptor (FcεRI), MCs express numerous G protein coupled receptors (GPCRs), which are the most common targets of drug therapy. Neurokinin 1 receptor (NK-1R) is expressed on MCs and contributes to IgE and non-IgE-mediated responses in mice. Although NK-1R antagonists are highly effective in modulating experimental allergic and inflammatory responses in mice they lack efficacy in humans. This article reviews recent findings that demonstrate that while neuropeptides (NPs) activate murine MCs via NK-1R and Mas related G protein coupled receptor B2 (MrgprB2), they activate human MCs via Mas-related G protein coupled receptor X2 (MRGPRX2). Interestingly, conventional NK-1R antagonists have off-target activity against mouse MrgprB2 but not human MRGPRX2. These findings suggest that the failure to translate studies with NK-1R antagonists from in vivo mouse studies to the clinic likely reflects their lack of effect on human MRGPRX2. A unique feature of MRGPRX2 that distinguishes it from other GPCRs is that it is activated by a diverse group of ligands that include; neuropeptides, cysteine proteases, antimicrobial peptides and cationic proteins released from activated eosinophils. Thus, the development of small molecule MRGPRX2-specific antagonists or neutralizing antibodies may provide new targets for the treatment of MC-mediated allergic and inflammatory diseases.
Keywords: MRGPRX2; Mast cells; MrgprB2; NK-1R; anaphylaxis; asthma.
Figures

References
-
- Gurish MF, Austen KF. Developmental origin and functional specialization of mast cell subsets. Immunity. 2012;37:25–33. - PubMed
-
- Irani AM, Bradford TR, Kepley CL, Schechter NM, Schwartz LB. Detection of MCT and MCTC types of human mast cells by immunohistochemistry using new monoclonal anti-tryptase and anti-chymase antibodies. J Histochem Cytochem. 1989;37:1509–1515. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
