SK channel enhancers attenuate Ca2+-dependent arrhythmia in hypertrophic hearts by regulating mito-ROS-dependent oxidation and activity of RyR
- PMID: 28096168
- PMCID: PMC5852621
- DOI: 10.1093/cvr/cvx005
SK channel enhancers attenuate Ca2+-dependent arrhythmia in hypertrophic hearts by regulating mito-ROS-dependent oxidation and activity of RyR
Abstract
Aims: Plasmamembrane small conductance Ca2+-activated K+ (SK) channels were implicated in ventricular arrhythmias in infarcted and failing hearts. Recently, SK channels were detected in the inner mitochondria membrane (IMM) (mSK), and their activation protected from acute ischaemia-reperfusion injury by reducing intracellular levels of reactive oxygen species (ROS). We hypothesized that mSK play an important role in regulating mitochondrial function in chronic cardiac diseases. We investigated the role of mSK channels in Ca2+-dependent ventricular arrhythmia using rat model of cardiac hypertrophy induced by banding of the ascending aorta thoracic aortic banding (TAB).
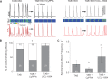
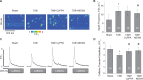
Methods and results: Dual Ca2+ and membrane potential optical mapping of whole hearts derived from TAB rats revealed that membrane-permeable SK enhancer NS309 (2 μM) improved aberrant Ca2+ homeostasis and abolished VT/VF induced by β-adrenergic stimulation. Using whole cell patch-clamp and confocal Ca2+ imaging of cardiomyocytes derived from TAB hearts (TCMs) we found that membrane-permeable SK enhancers NS309 and CyPPA (10 μM) attenuated frequency of spontaneous Ca2+ waves and delayed afterdepolarizations. Furthermore, mSK inhibition enhanced (UCL-1684, 1 μM); while activation reduced mitochondrial ROS production in TCMs measured with MitoSOX. Protein oxidation assays demonstrated that increased oxidation of ryanodine receptors (RyRs) in TCMs was reversed by SK enhancers. Experiments in permeabilized TCMs showed that SK enhancers restored SR Ca2+ content, suggestive of substantial improvement in RyR function.
Conclusion: These data suggest that enhancement of mSK channels in hypertrophic rat hearts protects from Ca2+-dependent arrhythmia and suggest that the protection is mediated via decreased mitochondrial ROS and subsequent decreased oxidation of reactive cysteines in RyR, which ultimately leads to stabilization of RyR-mediated Ca2+ release.
Keywords: Cardiac hypertrophy; Reactive oxygen species; Ryanodine receptor; Small conductance Ca2+-activated K+ channels; Ventricular arrhythmia.
Published on behalf of the European Society of Cardiology. All rights reserved. © The Author 2017. For permissions, please email: journals.permissions@oup.com.
Figures






References
-
- Zheng ZJ, Croft JB, Giles WH, Mensah GA.. Sudden cardiac death in the United States, 1989 to 1998. Circulation 2001;104:2158–2163. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

