Oxidized CaMKII promotes asthma through the activation of mast cells
- PMID: 28097237
- PMCID: PMC5214090
- DOI: 10.1172/jci.insight.90139
Oxidized CaMKII promotes asthma through the activation of mast cells
Abstract
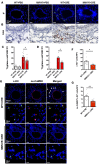
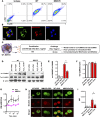
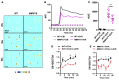
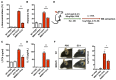
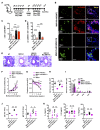
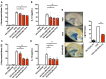
Oxidation of calmodulin-dependent protein kinase II (ox-CaMKII) by ROS has been associated with asthma. However, the contribution of ox-CaMKII to the development of asthma remains to be fully characterized. Here, we tested the effect of ox-CaMKII on IgE-mediated mast cell activation in an allergen-induced mouse model of asthma using oxidant-resistant CaMKII MMVVδ knockin (MMVVδ) mice. Compared with WT mice, the allergen-challenged MMVVδ mice displayed less airway hyperresponsiveness (AHR) and inflammation. These MMVVδ mice exhibited reduced levels of ROS and diminished recruitment of mast cells to the lungs. OVA-activated bone marrow-derived mast cells (BMMCs) from MMVVδ mice showed a significant inhibition of ROS and ox-CaMKII expression. ROS generation was dependent on intracellular Ca2+ concentration in BMMCs. Importantly, OVA-activated MMVVδ BMMCs had suppressed degranulation, histamine release, leukotriene C4, and IL-13 expression. Adoptive transfer of WT, but not MMVVδ, BMMCs, reversed the alleviated AHR and inflammation in allergen-challenged MMVVδ mice. The CaMKII inhibitor KN-93 significantly suppressed IgE-mediated mast cell activation and asthma. These studies support a critical but previously unrecognized role of ox-CaMKII in mast cells that promotes asthma and suggest that therapies to reduce ox-CaMKII may be a novel approach for asthma.
Conflict of interest statement
M.E. Anderson is a cofounder of Allosteros Therapeutics, a biotech company aiming to develop CaMKII-based therapies.
Figures


References
-
- Jarjour NN, Calhoun WJ. Enhanced production of oxygen radicals in asthma. J Lab Clin Med. 1994;123(1):131–136. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

