Splenic CD4+ T Cells in Progressive Visceral Leishmaniasis Show a Mixed Effector-Regulatory Phenotype and Impair Macrophage Effector Function through Inhibitory Receptor Expression
- PMID: 28103263
- PMCID: PMC5245871
- DOI: 10.1371/journal.pone.0169496
Splenic CD4+ T Cells in Progressive Visceral Leishmaniasis Show a Mixed Effector-Regulatory Phenotype and Impair Macrophage Effector Function through Inhibitory Receptor Expression
Abstract
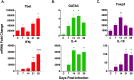
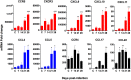
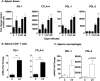
Visceral leishmaniasis (VL), caused by infection with the intracellular protozoan Leishmania donovani, is a chronic progressive disease with a relentlessly increasing parasite burden in the spleen, liver and bone marrow. The disease is characterized by fever, splenomegaly, cachexia, and pancytopenia, and progresses to death if not treated. Control of Leishmania infection is mediated by Th1 (IFNγ-producing) CD4+ T cells, which activate macrophages to produce nitric oxide and kill intracellular parasites. However, despite expansion of CD4+ T cells and increased IFNγ expression in the spleen, humans with active VL do not control the infection. We used an experimental model of chronic progressive VL in hamsters, which mimics clinical and pathological features seen in humans, to better understand the mechanisms that lead to progressive disease. Transcriptional profiling of the spleen during chronic infection revealed expression of markers of both T cell activation and inhibition. CD4+ T cells isolated from the spleen during chronic progressive VL showed mixed expression of Th1 and Th2 cytokines and chemokines, and were marginally effective in controlling infection in an ex vivo T cell-macrophage co-culture system. Splenic CD4+ T cells and macrophages from hamsters with VL showed increased expression of inhibitory receptors and their ligands, respectively. Blockade of the inhibitory receptor PD-L2 led to a significant decrease in parasite burden, revealing a pathogenic role for the PD-1 pathway in chronic VL. PD-L2 blockade was associated with a dramatic reduction in expression of host arginase 1, but no change in IFNγ and inducible nitric oxide synthase. Thus, the expression of counter-regulatory molecules on splenic CD4+ T cells and macrophages promotes a more permissive macrophage phenotype and attenuates intracellular parasite control in chronic progressive VL. Host-directed adjunctive therapy targeting the PD-1 regulatory pathway may be efficacious for VL.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





Similar articles
-
Transcriptional Profiling in Experimental Visceral Leishmaniasis Reveals a Broad Splenic Inflammatory Environment that Conditions Macrophages toward a Disease-Promoting Phenotype.PLoS Pathog. 2017 Jan 31;13(1):e1006165. doi: 10.1371/journal.ppat.1006165. eCollection 2017 Jan. PLoS Pathog. 2017. PMID: 28141856 Free PMC article.
-
The hamster as a model of human visceral leishmaniasis: progressive disease and impaired generation of nitric oxide in the face of a prominent Th1-like cytokine response.J Immunol. 2001 Feb 1;166(3):1912-20. doi: 10.4049/jimmunol.166.3.1912. J Immunol. 2001. PMID: 11160239
-
PD-1 function in apoptosis of T lymphocytes in canine visceral leishmaniasis.Immunobiology. 2016 Aug;221(8):879-88. doi: 10.1016/j.imbio.2016.03.007. Epub 2016 Mar 18. Immunobiology. 2016. PMID: 27016050
-
The role of CD4+ T cells in visceral leishmaniasis; new and emerging roles for NKG7 and TGFβ.Front Cell Infect Microbiol. 2024 May 31;14:1414493. doi: 10.3389/fcimb.2024.1414493. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 38881737 Free PMC article. Review.
-
Balancing immunity and pathology in visceral leishmaniasis.Immunol Cell Biol. 2007 Feb-Mar;85(2):138-47. doi: 10.1038/sj.icb7100011. Epub 2006 Dec 5. Immunol Cell Biol. 2007. PMID: 17146466 Review.
Cited by
-
Interleukin 2 is an Upstream Regulator of CD4+ T Cells From Visceral Leishmaniasis Patients With Therapeutic Potential.J Infect Dis. 2019 Jun 5;220(1):163-173. doi: 10.1093/infdis/jiz074. J Infect Dis. 2019. PMID: 30796820 Free PMC article.
-
A Calcium- and GTP-Dependent Transglutaminase in Leishmania infantum.Vet Sci. 2023 Mar 20;10(3):234. doi: 10.3390/vetsci10030234. Vet Sci. 2023. PMID: 36977273 Free PMC article.
-
Immunochemotherapy for visceral leishmaniasis: combinatorial action of Miltefosine plus LBSapMPL vaccine improves adaptative Th1 immune response with control of splenic parasitism in experimental hamster model.Parasitology. 2022 Mar;149(3):371-379. doi: 10.1017/S0031182021001906. Epub 2021 Nov 2. Parasitology. 2022. PMID: 35264268 Free PMC article.
-
Splenic macrophage functional profile and its role in the immunopathogenesis of canine visceral leishmaniasis.Front Immunol. 2025 Jun 20;16:1617751. doi: 10.3389/fimmu.2025.1617751. eCollection 2025. Front Immunol. 2025. PMID: 40621456 Free PMC article.
-
Transcriptome Analysis Identifies Immune Markers Related to Visceral Leishmaniasis Establishment in the Experimental Model of BALB/c Mice.Front Immunol. 2019 Nov 26;10:2749. doi: 10.3389/fimmu.2019.02749. eCollection 2019. Front Immunol. 2019. PMID: 31849951 Free PMC article.
References
-
- Finot MJ, Stenou MT, Guyot G, Cerf MA, Etienne G, Blech MF. [Enteric isolation in a normal room]. Soins. 1987;(507):III–IV. - PubMed
-
- Pearson RD, Sousa AdQ. Clinical spectrum of Leishmaniasis. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 1996;22(1). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

