Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice
- PMID: 28104796
- PMCID: PMC5542057
- DOI: 10.1126/science.aag1381
Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice
Abstract
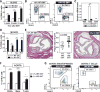
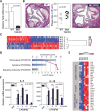
Human aging is associated with an increased frequency of somatic mutations in hematopoietic cells. Several of these recurrent mutations, including those in the gene encoding the epigenetic modifier enzyme TET2, promote expansion of the mutant blood cells. This clonal hematopoiesis correlates with an increased risk of atherosclerotic cardiovascular disease. We studied the effects of the expansion of Tet2-mutant cells in atherosclerosis-prone, low-density lipoprotein receptor-deficient (Ldlr-/-) mice. We found that partial bone marrow reconstitution with TET2-deficient cells was sufficient for their clonal expansion and led to a marked increase in atherosclerotic plaque size. TET2-deficient macrophages exhibited an increase in NLRP3 inflammasome-mediated interleukin-1β secretion. An NLRP3 inhibitor showed greater atheroprotective activity in chimeric mice reconstituted with TET2-deficient cells than in nonchimeric mice. These results support the hypothesis that somatic TET2 mutations in blood cells play a causal role in atherosclerosis.
Copyright © 2017, American Association for the Advancement of Science.
Figures


Comment in
-
Cardiovascular disease: Commonality with cancer.Nature. 2017 Mar 2;543(7643):45-47. doi: 10.1038/nature21505. Epub 2017 Feb 22. Nature. 2017. PMID: 28225756 No abstract available.
-
Hematopoietic stem cells gone rogue.Science. 2017 Feb 24;355(6327):798-799. doi: 10.1126/science.aam7939. Science. 2017. PMID: 28232539 No abstract available.
References
-
- Khot UN, Khot MB, Bajzer CT, Sapp SK, Ohman EM, Brener SJ, Ellis SG, Lincoff AM, Topol EJ. JAMA. 2003;290:898–904. - PubMed
-
- Fernández-Friera L, Peñalvo JL, Fernández-Ortiz A, Ibañez B, López-Melgar B, Laclaustra M, Oliva B, Mocoroa A, Mendiguren J, Martínez de Vega V, García L, Molina J, Sánchez-González J, Guzmán G, Alonso-Farto JC, Guallar E, Civeira F, Sillesen H, Pocock S, Ordovás JM, Sanz G, Jiménez-Borreguero LJ, Fuster V. Circulation. 2015;131:2104–2113. - PubMed
-
- Laclaustra M, Casasnovas JA, Fernández-Ortiz A, Fuster V, León-Latre M, Jiménez-Borreguero LJ, Pocovi M, Hurtado-Roca Y, Ordovas JM, Jarauta E, Guallar E, Ibañez B, Civeira F. J. Am. Coll. Cardiol. 2016;67:1263–1274. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

