Insulin-Like Growth Factor-1-Mediated DNA Repair in Irradiated Salivary Glands Is Sirtuin-1 Dependent
- PMID: 28106504
- PMCID: PMC5331616
- DOI: 10.1177/0022034516677529
Insulin-Like Growth Factor-1-Mediated DNA Repair in Irradiated Salivary Glands Is Sirtuin-1 Dependent
Abstract
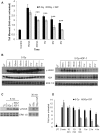
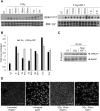
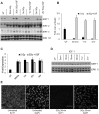
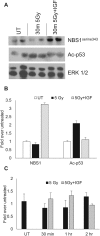
Ionizing radiation is one of the most common cancer treatments; however, the treatment leads to a wide range of debilitating side effects. In patients with head and neck cancer (HNC), the surrounding normal salivary gland is extremely sensitive to therapeutic radiation, and damage to this tissue results in various oral complications and decreased quality of life (QOL). In the current study, mice treated with targeted head and neck radiation showed a significant increase in double-stranded breaks (DSB) in the DNA of parotid salivary gland cells immediately after treatment, and this remained elevated 3 h posttreatment. In contrast, mice pretreated with insulin-like growth factor-1 (IGF-1) showed resolution of the same amount of initial DNA damage by 3 h posttreatment. At acute time points (30 min to 2 h), irradiated parotid glands had significantly decreased levels of the histone deactylase Sirtuin-1 (SirT-1) which has been previously shown to function in DNA repair. Pretreatment with IGF-1 increased SirT-1 protein levels and increased deacetylation of SirT-1 targets involved in DNA repair. Pharmacological inhibition of SirT-1 activity decreased the IGF-1-mediated resolution of DSB. These data suggest that IGF-1 promotes DNA repair in irradiated parotid glands through the maintenance and activation of SirT-1.
Keywords: DNA repair; IGF-1; Sirtuin-1; radiation; salivary glands; xerostomia.
Conflict of interest statement
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
Figures

Similar articles
-
Yap activation in irradiated parotid salivary glands is regulated by ROCK activity.PLoS One. 2020 Nov 5;15(11):e0232921. doi: 10.1371/journal.pone.0232921. eCollection 2020. PLoS One. 2020. PMID: 33151927 Free PMC article.
-
TAT-mediated delivery of Tousled protein to salivary glands protects against radiation-induced hypofunction.Int J Radiat Oncol Biol Phys. 2012 Sep 1;84(1):257-65. doi: 10.1016/j.ijrobp.2011.10.064. Epub 2012 Jan 26. Int J Radiat Oncol Biol Phys. 2012. PMID: 22285666
-
Restoration of radiation therapy-induced salivary gland dysfunction in mice by post therapy IGF-1 administration.BMC Cancer. 2010 Aug 10;10:417. doi: 10.1186/1471-2407-10-417. BMC Cancer. 2010. PMID: 20698985 Free PMC article.
-
Pathophysiology and management of radiation-induced xerostomia.J Support Oncol. 2005 May-Jun;3(3):191-200. J Support Oncol. 2005. PMID: 15915820 Review.
-
Transfer of the AQP1 cDNA for the correction of radiation-induced salivary hypofunction.Biochim Biophys Acta. 2006 Aug;1758(8):1071-7. doi: 10.1016/j.bbamem.2005.11.006. Epub 2005 Dec 5. Biochim Biophys Acta. 2006. PMID: 16368071 Review.
Cited by
-
Dual Characters of GH-IGF1 Signaling Pathways in Radiotherapy and Post-radiotherapy Repair of Cancers.Front Cell Dev Biol. 2021 Jun 9;9:671247. doi: 10.3389/fcell.2021.671247. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34178997 Free PMC article. Review.
-
Secretome Analysis of Inductive Signals for BM-MSC Transdifferentiation into Salivary Gland Progenitors.Int J Mol Sci. 2020 Nov 28;21(23):9055. doi: 10.3390/ijms21239055. Int J Mol Sci. 2020. PMID: 33260559 Free PMC article.
-
Growth hormone and radiation therapy: friend, foe, or both?Endocr Relat Cancer. 2024 Jan 24;31(3):e220371. doi: 10.1530/ERC-22-0371. Print 2024 Mar 1. Endocr Relat Cancer. 2024. PMID: 38174978 Free PMC article. Review.
-
Growth Hormone Induces Colon DNA Damage Independent of IGF-1.Endocrinology. 2019 Jun 1;160(6):1439-1447. doi: 10.1210/en.2019-00132. Endocrinology. 2019. PMID: 31002310 Free PMC article.
-
The single-strand DNA-binding protein SSB1 is involved in the expression of salivary gland radiation injury repair.Front Pharmacol. 2024 Oct 15;15:1471996. doi: 10.3389/fphar.2024.1471996. eCollection 2024. Front Pharmacol. 2024. PMID: 39474611 Free PMC article.
References
-
- Al-Ejeh F, Kumar R, Wiegmans A, Lakhani SR, Brown MP, Khanna KK. 2010. Harnessing the complexity of DNA-damage response pathways to improve cancer treatment outcomes. Oncogene. 29(46):6085–6098. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

