Neuroprotective Strategy in Retinal Degeneration: Suppressing ER Stress-Induced Cell Death via Inhibition of the mTOR Signal
- PMID: 28106827
- PMCID: PMC5297831
- DOI: 10.3390/ijms18010201
Neuroprotective Strategy in Retinal Degeneration: Suppressing ER Stress-Induced Cell Death via Inhibition of the mTOR Signal
Abstract
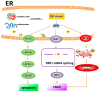
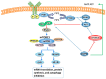
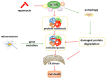
The retina is a specialized sensory organ, which is essential for light detection and visual formation in the human eye. Inherited retinal degenerations are a heterogeneous group of eye diseases that can eventually cause permanent vision loss. UPR (unfolded protein response) and ER (endoplasmic reticulum) stress plays an important role in the pathological mechanism of retinal degenerative diseases. mTOR (the mammalian target of rapamycin) kinase, as a signaling hub, controls many cellular processes, covering protein synthesis, RNA translation, ER stress, and apoptosis. Here, the hypothesis that inhibition of mTOR signaling suppresses ER stress-induced cell death in retinal degenerative disorders is discussed. This review surveys knowledge of the influence of mTOR signaling on ER stress arising from misfolded proteins and genetic mutations in retinal degenerative diseases and highlights potential neuroprotective strategies for treatment and therapeutic implications.
Keywords: ER stress; apoptosis; mTOR; retinal degeneration; retinal neuroprotection; unfolded protein response.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Bidirectional crosstalk between endoplasmic reticulum stress and mTOR signaling.Trends Cell Biol. 2012 May;22(5):274-82. doi: 10.1016/j.tcb.2012.02.006. Epub 2012 Mar 21. Trends Cell Biol. 2012. PMID: 22444729 Review.
-
Rapamycin attenuates visible light-induced injury in retinal photoreceptor cells via inhibiting endoplasmic reticulum stress.Brain Res. 2014 May 14;1563:1-12. doi: 10.1016/j.brainres.2014.02.020. Epub 2014 Mar 4. Brain Res. 2014. PMID: 24607296
-
The GST-BHMT assay reveals a distinct mechanism underlying proteasome inhibition-induced macroautophagy in mammalian cells.Autophagy. 2015;11(5):812-32. doi: 10.1080/15548627.2015.1034402. Autophagy. 2015. PMID: 25984893 Free PMC article.
-
Bridges between mitochondrial oxidative stress, ER stress and mTOR signaling in pancreatic β cells.Cell Signal. 2016 Aug;28(8):1099-104. doi: 10.1016/j.cellsig.2016.05.007. Epub 2016 May 14. Cell Signal. 2016. PMID: 27185188 Review.
-
Algae Undaria pinnatifida Protects Hypothalamic Neurons against Endoplasmic Reticulum Stress through Akt/mTOR Signaling.Molecules. 2015 Nov 25;20(12):20998-1009. doi: 10.3390/molecules201219744. Molecules. 2015. PMID: 26610463 Free PMC article.
Cited by
-
Retinal Inflammation, Cell Death and Inherited Retinal Dystrophies.Int J Mol Sci. 2021 Feb 20;22(4):2096. doi: 10.3390/ijms22042096. Int J Mol Sci. 2021. PMID: 33672611 Free PMC article. Review.
-
Branched-chain Amino Acids: Catabolism in Skeletal Muscle and Implications for Muscle and Whole-body Metabolism.Front Physiol. 2021 Jul 20;12:702826. doi: 10.3389/fphys.2021.702826. eCollection 2021. Front Physiol. 2021. PMID: 34354601 Free PMC article. Review.
-
Induction of Heat Shock Protein 70 in Mouse RPE as an In Vivo Model of Transpupillary Thermal Stimulation.Int J Mol Sci. 2020 Mar 17;21(6):2063. doi: 10.3390/ijms21062063. Int J Mol Sci. 2020. PMID: 32192227 Free PMC article.
-
Are mTOR and Endoplasmic Reticulum Stress Pathway Genes Associated with Oral and Bone Diseases?Caries Res. 2019;53(3):235-241. doi: 10.1159/000492675. Epub 2018 Sep 11. Caries Res. 2019. PMID: 30205378 Free PMC article.
-
Disturbance of neuron-microglia crosstalk mediated by GRP78 in Neuropsychiatric systemic lupus erythematosus mice.J Neuroinflammation. 2023 Jun 26;20(1):150. doi: 10.1186/s12974-023-02832-8. J Neuroinflammation. 2023. PMID: 37365565 Free PMC article.
References
-
- Kolb H., Nelson R. The Organization of Photoreceptor to Bipolar Synapses in the Outer Plexiform Layer. Springer; Houten, The Netherlands: 1995. pp. 273–296.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

