Cytochrome c Oxidase Biogenesis and Metallochaperone Interactions: Steps in the Assembly Pathway of a Bacterial Complex
- PMID: 28107462
- PMCID: PMC5249081
- DOI: 10.1371/journal.pone.0170037
Cytochrome c Oxidase Biogenesis and Metallochaperone Interactions: Steps in the Assembly Pathway of a Bacterial Complex
Abstract
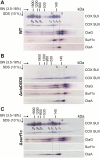
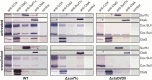
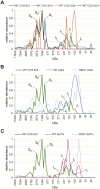
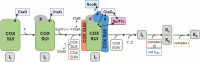
Biogenesis of mitochondrial cytochrome c oxidase (COX) is a complex process involving the coordinate expression and assembly of numerous subunits (SU) of dual genetic origin. Moreover, several auxiliary factors are required to recruit and insert the redox-active metal compounds, which in most cases are buried in their protein scaffold deep inside the membrane. Here we used a combination of gel electrophoresis and pull-down assay techniques in conjunction with immunostaining as well as complexome profiling to identify and analyze the composition of assembly intermediates in solubilized membranes of the bacterium Paracoccus denitrificans. Our results show that the central SUI passes through at least three intermediate complexes with distinct subunit and cofactor composition before formation of the holoenzyme and its subsequent integration into supercomplexes. We propose a model for COX biogenesis in which maturation of newly translated COX SUI is initially assisted by CtaG, a chaperone implicated in CuB site metallation, followed by the interaction with the heme chaperone Surf1c to populate the redox-active metal-heme centers in SUI. Only then the remaining smaller subunits are recruited to form the mature enzyme which ultimately associates with respiratory complexes I and III into supercomplexes.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Deciphering protein-protein interactions during the biogenesis of cytochrome c oxidase from Paracoccus denitrificans.FEBS J. 2015 Feb;282(3):537-49. doi: 10.1111/febs.13160. Epub 2014 Dec 15. FEBS J. 2015. PMID: 25420759
-
Protein chaperones mediating copper insertion into the CuA site of the aa3-type cytochrome c oxidase of Paracoccus denitrificans.Biochim Biophys Acta. 2015 Feb;1847(2):202-211. doi: 10.1016/j.bbabio.2014.11.001. Epub 2014 Nov 13. Biochim Biophys Acta. 2015. PMID: 25445316
-
Characterization of heme-binding properties of Paracoccus denitrificans Surf1 proteins.FEBS J. 2011 May;278(10):1769-78. doi: 10.1111/j.1742-4658.2011.08101.x. Epub 2011 Apr 11. FEBS J. 2011. PMID: 21418525
-
Role of Surf1 in heme recruitment for bacterial COX biogenesis.Biochim Biophys Acta. 2012 Jun;1817(6):928-37. doi: 10.1016/j.bbabio.2011.09.007. Epub 2011 Sep 16. Biochim Biophys Acta. 2012. PMID: 21945856 Review.
-
Biochemistry of Copper Site Assembly in Heme-Copper Oxidases: A Theme with Variations.Int J Mol Sci. 2019 Aug 5;20(15):3830. doi: 10.3390/ijms20153830. Int J Mol Sci. 2019. PMID: 31387303 Free PMC article. Review.
Cited by
-
Complexome Profiling: Assembly and Remodeling of Protein Complexes.Int J Mol Sci. 2021 Jul 21;22(15):7809. doi: 10.3390/ijms22157809. Int J Mol Sci. 2021. PMID: 34360575 Free PMC article. Review.
-
The Complexome of Dehalococcoides mccartyi Reveals Its Organohalide Respiration-Complex Is Modular.Front Microbiol. 2018 Jun 12;9:1130. doi: 10.3389/fmicb.2018.01130. eCollection 2018. Front Microbiol. 2018. PMID: 29946299 Free PMC article.
-
The World of Stable Ribonucleoproteins and Its Mapping With Grad-Seq and Related Approaches.Front Mol Biosci. 2021 Apr 7;8:661448. doi: 10.3389/fmolb.2021.661448. eCollection 2021. Front Mol Biosci. 2021. PMID: 33898526 Free PMC article. Review.
-
Diversity of Cytochrome c Oxidase Assembly Proteins in Bacteria.Microorganisms. 2022 Apr 28;10(5):926. doi: 10.3390/microorganisms10050926. Microorganisms. 2022. PMID: 35630371 Free PMC article. Review.
-
Widespread Distribution and Functional Specificity of the Copper Importer CcoA: Distinct Cu Uptake Routes for Bacterial Cytochrome c Oxidases.mBio. 2018 Feb 27;9(1):e00065-18. doi: 10.1128/mBio.00065-18. mBio. 2018. PMID: 29487231 Free PMC article.
References
-
- Richter O-MH, Ludwig B. Heme Proteins, Cytochrome c Oxidase Encyclopedia of Metalloproteins. Springer: Kretsinger Robert H.; Uversky Vladimir N.; Permyakov Eugene A. (Eds.); 2013. p. 939–44.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

