Nanobody-based enzyme immunoassay for ochratoxin A in cereal with high resistance to matrix interference
- PMID: 28107910
- PMCID: PMC5862147
- DOI: 10.1016/j.talanta.2016.11.039
Nanobody-based enzyme immunoassay for ochratoxin A in cereal with high resistance to matrix interference
Abstract
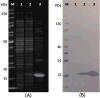
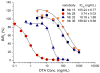
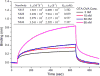
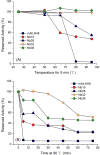
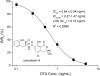
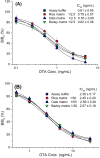
A sensitive indirect competitive nanobody-based enzyme linked immunosorbent assay (Nb-ELISA) for ochratoxin A (OTA) with high resistance to cereal matrix interference was developed. Nanobodies against OTA (Nb15, Nb28, Nb32, Nb36) were expressed in E. coli cells and their thermal stabilities were compared with that of an OTA-specific monoclonal antibody 6H8. All nanobodies could still retain their antigen-binding activity after exposure to temperature 95°C for 5min or to 90°C for 75min. Nb28 that exhibited the highest sensitivity in ELISA was selected for further research. An indirect competitive ELISA based on Nb28 was developed for OTA, with an IC50 of 0.64ng/mL and a linear range (IC20-IC80) of 0.27-1.47ng/mL. Cereal samples were analyzed following a 2.5 fold dilution of sample extracts, showing the good resistance to matrix interference of the Nb-ELSIA. The recovery of spiked cereal samples (rice, oats, barley) ranged from 80% to 105% and the Nb-ELISA results of OTA content in naturally contamined samples were in good agreement with those determined by a commercial ELISA kit. The results indicated the reliablity of nanobody as a promising immunoassay reagent for detection of mycotoxins in food matrix and its potential in biosensor development.
Keywords: Cereal; Immunoassay; Matrix interference; Nanobody; Ochratoxin A.
Copyright © 2016 Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Development of a Nanobody-AviTag Fusion Protein and Its Application in a Streptavidin-Biotin-Amplified Enzyme-Linked Immunosorbent Assay for Ochratoxin A in Cereal.Anal Chem. 2018 Sep 4;90(17):10628-10634. doi: 10.1021/acs.analchem.8b03085. Epub 2018 Aug 22. Anal Chem. 2018. PMID: 30092629 Free PMC article.
-
Nanobody-Alkaline Phosphatase Fusion Protein-Based Enzyme-Linked Immunosorbent Assay for One-Step Detection of Ochratoxin A in Rice.Sensors (Basel). 2018 Nov 20;18(11):4044. doi: 10.3390/s18114044. Sensors (Basel). 2018. PMID: 30463338 Free PMC article.
-
Development of a nanobody-alkaline phosphatase fusion protein and its application in a highly sensitive direct competitive fluorescence enzyme immunoassay for detection of ochratoxin A in cereal.Anal Chem. 2015 Jan 20;87(2):1387-94. doi: 10.1021/ac504305z. Epub 2015 Jan 6. Anal Chem. 2015. PMID: 25531426 Free PMC article.
-
Ochratoxin A and ochratoxin-producing fungi on cereal grain in China: a review.Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2015;32(4):461-70. doi: 10.1080/19440049.2014.996787. Epub 2015 Jan 9. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2015. PMID: 25571918 Review.
-
Anti-idiotypic nanobody: A strategy for development of sensitive and green immunoassay for Fumonisin B₁.Talanta. 2015 Oct 1;143:388-393. doi: 10.1016/j.talanta.2015.05.010. Epub 2015 May 11. Talanta. 2015. PMID: 26078175 Review.
Cited by
-
NANOBODIES®: A Review of Diagnostic and Therapeutic Applications.Int J Mol Sci. 2023 Mar 22;24(6):5994. doi: 10.3390/ijms24065994. Int J Mol Sci. 2023. PMID: 36983063 Free PMC article. Review.
-
A Chemiluminescence Enzyme Immunoassay Based on Biotinylated Nanobody and Streptavidin Amplification for Diazinon Sensitive Quantification.Biosensors (Basel). 2023 May 25;13(6):577. doi: 10.3390/bios13060577. Biosensors (Basel). 2023. PMID: 37366942 Free PMC article.
-
Nanobody-based indirect competitive ELISA for the detection of aflatoxin M1 in dairy products.Sci Rep. 2025 Jan 4;15(1):785. doi: 10.1038/s41598-024-83869-4. Sci Rep. 2025. PMID: 39755714 Free PMC article.
-
Intelligent Electrochemical Point-of-Care Test Method with Interface Control Based on DNA Pyramids: Aflatoxin B1 Detection in Food and the Environment.Foods. 2023 Dec 12;12(24):4447. doi: 10.3390/foods12244447. Foods. 2023. PMID: 38137251 Free PMC article.
-
Development of a Nanobody-AviTag Fusion Protein and Its Application in a Streptavidin-Biotin-Amplified Enzyme-Linked Immunosorbent Assay for Ochratoxin A in Cereal.Anal Chem. 2018 Sep 4;90(17):10628-10634. doi: 10.1021/acs.analchem.8b03085. Epub 2018 Aug 22. Anal Chem. 2018. PMID: 30092629 Free PMC article.
References
-
- Yu FY, Vdovenko MM, Wang JJ, Sakharov IY. J Agri Food Chem. 2011;59:809–813. - PubMed
-
- Liu X, Xu Y, He Q, He Z, Xiong Z. J Agri Food Chem. 2013;61:4765–4770. - PubMed
-
- Min WK, Kim SG, H J. Seo, Food Chem. 2015;188:604–611. - PubMed
-
- Arola HO, Tullila A, Kiljunen H, Campbell K, Siitari H, Nevanen TK. Anal Chem. 2016;88:2446–2452. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

