Mutations of two acidic residues at the cytoplasmic end of segment IIIS6 of an insect sodium channel have distinct effects on pyrethroid resistance
- PMID: 28111191
- PMCID: PMC5730327
- DOI: 10.1016/j.ibmb.2017.01.007
Mutations of two acidic residues at the cytoplasmic end of segment IIIS6 of an insect sodium channel have distinct effects on pyrethroid resistance
Abstract
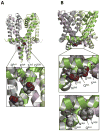
Mutations in sodium channels are known to confer knockdown resistance to pyrethroid insecticides, such as permethrin and cypermethrin, in various agricultural pests and disease vectors. Double mutations, D3i28V and E3i32G, were detected in cypermethrin-resistant Helicoverpa armigera and Heliothis virescens populations. However, the role of the two mutations in pyrethroid resistance remains unclear. In this study, we introduced the mutations into the cockroach sodium channel, BgNav1-1a, and examined their effects on channel gating and pyrethroid sensitivity in Xenopus oocytes. D3i28V alone and the double mutation, D3i28V/E3i32G, shifted the voltage dependence of activation in the depolarizing direction by 15 mV and 20 mV, respectively, whereas E3i32G had no significant effect. D3i28V reduced the amplitude of tail currents induced by permethrin and NRDC 157 (Type I pyrethroids) and deltamethrin and cypermethrin (Type II pyrethroids), whereas E3i32G alone had no effect. Intriguingly, the amplitude of Type II pyrethroid-induced tail current from D3i28V/E3i32G channels was similar to that of BgNav1-1a channels, but the decay of the tail currents was accelerated. Such effects were not observed for Type I pyrethroid-induced tail currents. Computational analysis based on the model of dual pyrethroid receptors on insect sodium channels predicted D3i28V and E3i32G exert their effects on channel gating and pyrethroid action via allosteric mechanisms. Our results not only illustrate the distinct effect of the D3i28V/E3i32G double mutations on Type I vs. Type II pyrethroids, but also reinforce the concept that accelerated decay of tail currents can be an effective mechanism of pyrethroid resistance to Type II pyrethroids.
Keywords: Knockdown resistance; Pyrethroid receptor; Sodium channel; Type I pyrethroids; Type II pyrethroids.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures

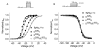


Similar articles
-
Role of mutation G255A in modulating pyrethroid sensitivity in insect sodium channels.Int J Biol Macromol. 2025 Mar;295:139455. doi: 10.1016/j.ijbiomac.2025.139455. Epub 2025 Jan 6. Int J Biol Macromol. 2025. PMID: 39778843
-
Alanine to valine substitutions in the pore helix IIIP1 and linker-helix IIIL45 confer cockroach sodium channel resistance to DDT and pyrethroids.Neurotoxicology. 2017 May;60:197-206. doi: 10.1016/j.neuro.2016.06.009. Epub 2016 Jun 18. Neurotoxicology. 2017. PMID: 27328896 Free PMC article.
-
A sodium channel mutation identified in Aedes aegypti selectively reduces cockroach sodium channel sensitivity to type I, but not type II pyrethroids.Insect Biochem Mol Biol. 2011 Jan;41(1):9-13. doi: 10.1016/j.ibmb.2010.09.005. Epub 2010 Sep 29. Insect Biochem Mol Biol. 2011. PMID: 20869441 Free PMC article.
-
The molecular interactions of pyrethroid insecticides with insect and mammalian sodium channels.Pest Manag Sci. 2001 Oct;57(10):877-88. doi: 10.1002/ps.392. Pest Manag Sci. 2001. PMID: 11695180 Review.
-
Poisoning due to pyrethroids.Toxicol Rev. 2005;24(2):93-106. doi: 10.2165/00139709-200524020-00003. Toxicol Rev. 2005. PMID: 16180929 Review.
Cited by
-
Vgsc-interacting proteins are genetically associated with pyrethroid resistance in Aedes aegypti.PLoS One. 2019 Jan 29;14(1):e0211497. doi: 10.1371/journal.pone.0211497. eCollection 2019. PLoS One. 2019. PMID: 30695054 Free PMC article.
-
Pyrethroids in an AlphaFold2 Model of the Insect Sodium Channel.Insects. 2022 Aug 18;13(8):745. doi: 10.3390/insects13080745. Insects. 2022. PMID: 36005370 Free PMC article.
References
-
- Ahuja S, Mukund S, Deng L, Khakh K, Chang E, Ho H, Shriver S, Young C, Lin S, Johnson JP, Jr, Wu P, Li J, Coons M, Tam C, Brillantes B, Sampang H, Mortara K, Bowman KK, Clark KR, Estevez A, Xie Z, Verschoof H, Grimwood M, Dehnhardt C, Andrez JC, Focken T, Sutherlin DP, Safina BS, Starovasnik MA, Ortwine DF, Franke Y, Cohen CJ, Hackos DH, Koth CM, Payandeh J. Structural basis of Nav1.7 inhibition by an isoform-selective small-molecule antagonist. Science. 2015;350:aac5464. - PubMed
-
- Bloomquist JR. Ion channels as targets for insecticides. Annu Rev Entomol. 1996;41:163–190. - PubMed
-
- Bloomquist JR, Soderlund DM. Pyrethroid insecticides and DDT modify alkaloid-dependent sodium channel activation and its enhancement by sea anemone toxin. Mol Pharmacol. 1988;33:543–550. - PubMed
-
- Catterall WA, Cestele S, Yarov-Yarovoy V, Yu FH, Konoki K, Scheuer T. Voltage-gated ion channels and gating modifier toxins. Toxicon. 2007;49:124–141. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

