Close contact interferon-gamma response to the new PstS1(285-374):CPF10: a preliminary 1-year follow-up study
- PMID: 28114976
- PMCID: PMC5259914
- DOI: 10.1186/s13104-016-2360-4
Close contact interferon-gamma response to the new PstS1(285-374):CPF10: a preliminary 1-year follow-up study
Abstract
Background: The available diagnostic tools for latent tuberculosis (TB) infection (LTBI) via interferon-gamma (IFN-g) release assays (IGRA) are based on ESAT6:CFP10 stimulation. However, the mycobacterial antigen PstS1 is also highly immunogenic and some of its fragments, such as PstS1(285-374), have shown higher immunoreactivity in LTBI than in active TB. PstS1(285-374), therefore, could increase the accuracy of the existing IGRA to detect LTBI. Thus, a new chimeric protein has recently been developed (PstS1(285-374):CFP10) showing potential for LTBI screening of recent close contacts (rCt) exposed to Mycobacterium tuberculosis. The aim of this study was to analyze the PstS1(285-374):CFP10 longitudinal IFN-g profile in comparison to ESAT6:CFP10 and full PstS1/CFP10 stimulation in a rCt cohort and correlate the responses to these in-house IGRA with any clinical changes/interventions that might occur.
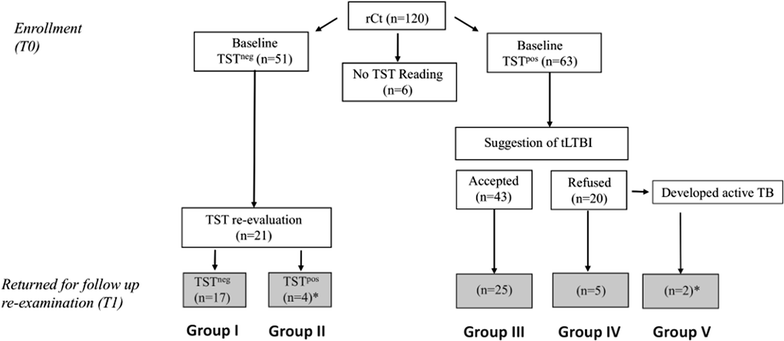
Methods: A free-of-cost, one-year follow up was offered to 120 rCt recruited in Rio de Janeiro, RJ, Brazil. Whole blood short-term (WBA), long-term stimulation (LSA) assays, and the tuberculin skin test (TST) were performed during follow up.
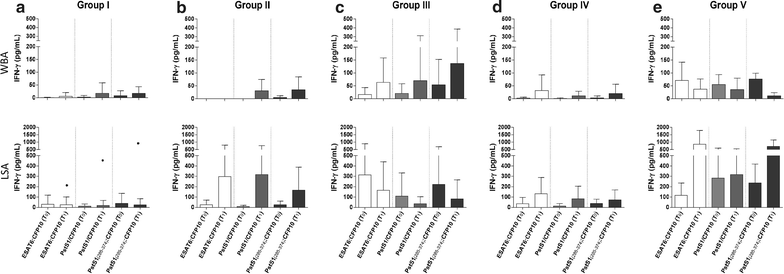
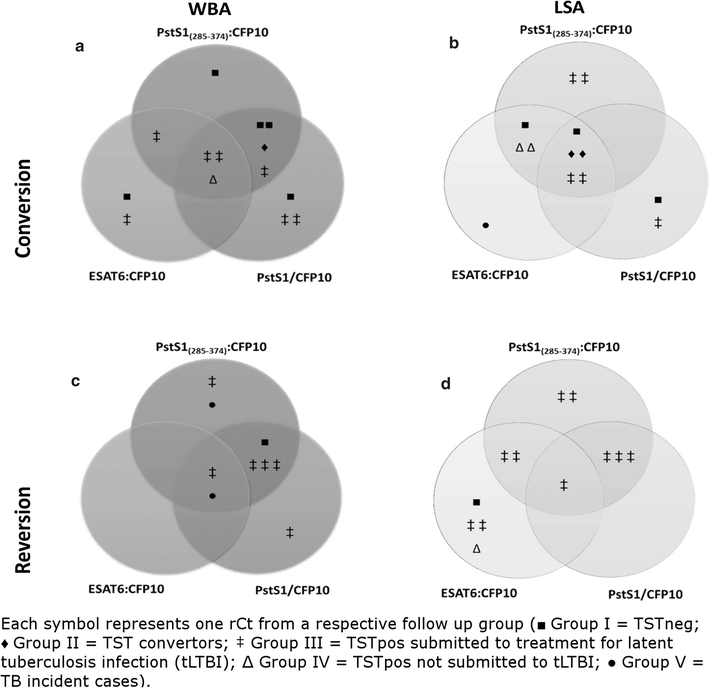
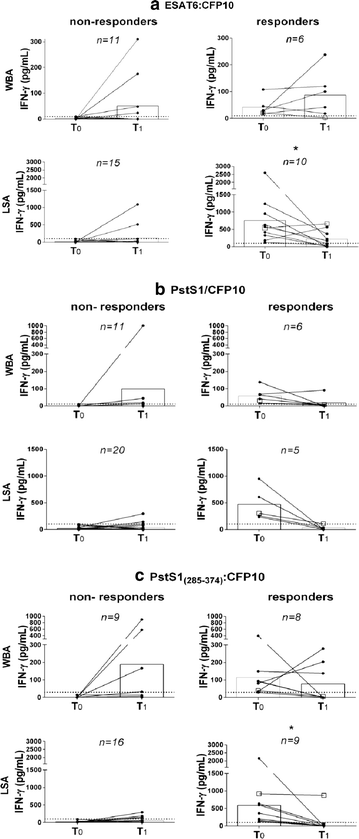
Results: Among the enrolled rCt, 44.2% (53/120) returned for re-evaluation and the control group (TST negative, n = 17) showed low IFN-g reactivity to all antigen stimulations during the entire follow up, except for one participant who had shown radiological evidence of past TB/LTBI. Both incident cases were detected by IGRA-PstS1(285-374):CFP10 during LTBI and after disease progression. Moreover, subsequent to the prophylactic treatment for LTBI (tLTBI), a significant regression in the LSA response was predominantly observed through stimulation of the new chimeric protein (8/10, 80%) followed by ESAT6:CFP10 (5/10, 50%) and PstS1/CFP10 (4/10, 40%). No clinical or epidemiological characteristics were exclusively shared among IGRA convertors.
Conclusion: It was demonstrated that the TST negative rCt without radiological evidence of LTBI/TB did not develop an IGRA-PstS1(285-374):CFP10 response during the one-year follow up. Moreover, all incident cases were detected by our new IGRA; and a significant decrement of LSA-PstS1(285-374):CFP10 reactivity post-prophylactic tLTBI was found. To our knowledge, this is the first study to monitor changes in the immune response profile of IGRA-PstS1(285-374):CFP10 among rCt during a consecutive one-year period, thus providing additional evidence of its potential in the detection of LTBI.
Keywords: Follow up; Interferon-gamma; LTBI; M. tuberculosis; Tuberculosis.
Figures
References
-
- Implementing the WHO Stop TB Strategy: a handbook for national tuberculosis control programmes. Geneva: World Health Organization; 2008. - PubMed
-
- Steffen RE, Caetano R, Pinto M, Chaves D, Ferrari R, Bastos M, de Abreu ST, Menzies D, Trajman A. Cost-effectiveness of quantiferon®-TB gold-in-tube versus tuberculin skin testing for contact screening and treatment of latent tuberculosis infection in Brazil. PLoS ONE. 2013;8(4):e59546. doi: 10.1371/journal.pone.0059546. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

