Osmotolerance in Escherichia coli Is Improved by Activation of Copper Efflux Genes or Supplementation with Sulfur-Containing Amino Acids
- PMID: 28115377
- PMCID: PMC5359503
- DOI: 10.1128/AEM.03050-16
Osmotolerance in Escherichia coli Is Improved by Activation of Copper Efflux Genes or Supplementation with Sulfur-Containing Amino Acids
Abstract
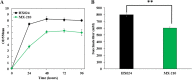
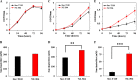
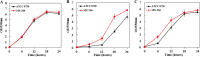
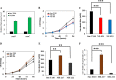
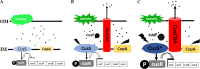
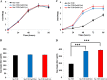
Improvement in the osmotolerance of Escherichia coli is essential for the production of high titers of various bioproducts. In this work, a cusS mutation that was identified in the previously constructed high-succinate-producing E. coli strain HX024 was investigated for its effect on osmotolerance. CusS is part of the two-component system CusSR that protects cells from Ag(I) and Cu(I) toxicity. Changing cusS from strain HX024 back to its original sequence led to a 24% decrease in cell mass and succinate titer under osmotic stress (12% glucose). When cultivated with a high initial glucose concentration (12%), introduction of the cusS mutation into parental strain Suc-T110 led to a 21% increase in cell mass and a 40% increase in succinate titer. When the medium was supplemented with 30 g/liter disodium succinate, the cusS mutation led to a 120% increase in cell mass and a 492% increase in succinate titer. Introducing the cusS mutation into the wild-type strain ATCC 8739 led to increases in cell mass of 87% with 20% glucose and 36% using 30 g/liter disodium succinate. The cusS mutation increased the expression of cusCFBA, and gene expression levels were found to be positively related to osmotolerance abilities. Because high osmotic stress has been associated with deleterious accumulation of Cu(I) in the periplasm, activation of CusCFBA may alleviate this effect by transporting Cu(I) out of the cells. This hypothesis was confirmed by supplementing sulfur-containing amino acids that can chelate Cu(I). Adding methionine or cysteine to the medium increased the osmotolerance of E. coli under anaerobic conditions.IMPORTANCE In this work, an activating Cus copper efflux system was found to increase the osmotolerance of E. coli In addition, new osmoprotectants were identified. Supplementation with methionine or cysteine led to an increase in osmotolerance of E. coli under anaerobic conditions. These new strategies for improving osmotolerance will be useful for improving the production of chemicals in industrial bioprocesses.
Keywords: Cu(I); CusS; Escherichia coli; copper efflux; methionine; osmotolerance.
Copyright © 2017 American Society for Microbiology.
Figures

Similar articles
-
A novel point mutation in RpoB improves osmotolerance and succinic acid production in Escherichia coli.BMC Biotechnol. 2017 Feb 13;17(1):10. doi: 10.1186/s12896-017-0337-6. BMC Biotechnol. 2017. PMID: 28193207 Free PMC article.
-
Regulation of Cu(I)/Ag(I) efflux genes in Escherichia coli by the sensor kinase CusS.FEMS Microbiol Lett. 2012 May;330(1):30-7. doi: 10.1111/j.1574-6968.2012.02529.x. Epub 2012 Mar 12. FEMS Microbiol Lett. 2012. PMID: 22348296 Free PMC article.
-
Copper efflux is induced during anaerobic amino acid limitation in Escherichia coli to protect iron-sulfur cluster enzymes and biogenesis.J Bacteriol. 2013 Oct;195(20):4556-68. doi: 10.1128/JB.00543-13. Epub 2013 Jul 26. J Bacteriol. 2013. PMID: 23893112 Free PMC article.
-
Metal export by CusCFBA, the periplasmic Cu(I)/Ag(I) transport system of Escherichia coli.Curr Top Membr. 2012;69:163-96. doi: 10.1016/B978-0-12-394390-3.00007-0. Curr Top Membr. 2012. PMID: 23046651 Review.
-
Escherichia coli mechanisms of copper homeostasis in a changing environment.FEMS Microbiol Rev. 2003 Jun;27(2-3):197-213. doi: 10.1016/S0168-6445(03)00049-4. FEMS Microbiol Rev. 2003. PMID: 12829268 Review.
Cited by
-
An economically and environmentally acceptable synthesis of chiral drug intermediate L-pipecolic acid from biomass-derived lysine via artificially engineered microbes.J Ind Microbiol Biotechnol. 2018 Jun;45(6):405-415. doi: 10.1007/s10295-018-2044-2. Epub 2018 May 10. J Ind Microbiol Biotechnol. 2018. PMID: 29749580
-
Soft-metal(loid)s induce protein aggregation in Escherichia coli.Front Microbiol. 2023 Nov 22;14:1281058. doi: 10.3389/fmicb.2023.1281058. eCollection 2023. Front Microbiol. 2023. PMID: 38075883 Free PMC article.
-
Recent advances in producing food additive L-malate: Chassis, substrate, pathway, fermentation regulation and application.Microb Biotechnol. 2023 Apr;16(4):709-725. doi: 10.1111/1751-7915.14206. Epub 2023 Jan 5. Microb Biotechnol. 2023. PMID: 36604311 Free PMC article. Review.
-
Multiomic Fermentation Using Chemically Defined Synthetic Hydrolyzates Revealed Multiple Effects of Lignocellulose-Derived Inhibitors on Cell Physiology and Xylose Utilization in Zymomonas mobilis.Front Microbiol. 2019 Nov 7;10:2596. doi: 10.3389/fmicb.2019.02596. eCollection 2019. Front Microbiol. 2019. PMID: 31787963 Free PMC article.
-
Assessing the Involvement of Selected Phenotypes of Pseudomonas simiae PICF7 in Olive Root Colonization and Biological Control of Verticillium dahliae.Plants (Basel). 2021 Feb 23;10(2):412. doi: 10.3390/plants10020412. Plants (Basel). 2021. PMID: 33672351 Free PMC article.
References
-
- Winkler JD, Garcia C, Olson M, Callaway E, Kao KC. 2014. Evolved osmotolerant Escherichia coli mutants frequently exhibit defective N-acetylglucosamine catabolism and point mutations in cell shape-regulating protein MreB. Appl Environ Microbiol 80:3729–3740. doi: 10.1128/AEM.00499-14. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

