Early Detection of Acute Drug-Induced Liver Injury in Mice by Noninvasive Near-Infrared Fluorescence Imaging
- PMID: 28115551
- PMCID: PMC5363778
- DOI: 10.1124/jpet.116.238378
Early Detection of Acute Drug-Induced Liver Injury in Mice by Noninvasive Near-Infrared Fluorescence Imaging
Abstract
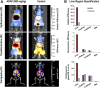
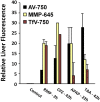
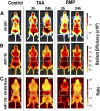
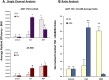
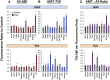
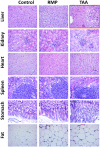
Hepatocellular and cholestatic forms of drug-induced liver injury (DILI) are major reasons for late-stage termination of small-molecule drug discovery research projects. Biochemical serum markers are limited in their ability to sensitively and specifically detect both of these common DILI forms in preclinical models, and tissue-specific approaches to assessing this are labor intensive, requiring extensive animal dosing, tissue preparation, and pathology assessment. In vivo fluorescent imaging offers noninvasive detection of biologic changes detected directly in the livers of living animals. Three different near-infrared fluorescent imaging probes, specific for cell death (Annexin-Vivo 750), matrix metalloproteases (MMPSense 750 FAST), and transferrin receptor (Transferrin-Vivo 750) were used to measure the effects of single bolus intraperitoneal doses of four different chemical agents known to induce liver injury. Hepatocellular injury-inducing agents, thioacetamide and acetaminophen, showed optimal injury detection with probe injection at 18-24 hours, the liver cholestasis-inducing drug rifampicin required early probe injection (2 hours), and chlorpromazine, which induces mixed hepatocellular/cholestatic injury, showed injury with both early and late injection. Different patterns of liver responses were seen among these different imaging probes, and no one probe detected injury by all four compounds. By using a cocktail of these three near-infrared fluorescent imaging probes, all labeled with 750-nm fluorophores, each of the four different DILI agents induced comparable tissue injury within the liver region, as assessed by epifluorescence imaging. A strategy of probe cocktail injection in separate cohorts at 2 hours and at 20-24 hours allowed the effective detection of drugs with either early- or late-onset injury.
Copyright © 2017 by The Author(s).
Figures




References
-
- Ackerman Z, Pappo O, Link G, Glazer M, Grozovski M. (2015) Liver toxicity of thioacetamide is increased by hepatocellular iron overload. Biol Trace Elem Res 163:169–176. - PubMed
-
- Ahuja V, Sharma S. (2014) Drug safety testing paradigm, current progress and future challenges: an overview. J Appl Toxicol 34:576–594. - PubMed
-
- al-Bader A, Mathew TC, Khoursheed M, Asfar S, al-Sayer H, Dashti HM. (2000) Thioacetamide toxicity and the spleen: histological and biochemical analysis. Anat Histol Embryol 29:3–8. - PubMed
-
- Al-Olayan EM, El-Khadragy MF, Aref AM, Othman MS, Kassab RB, Abdel Moneim AE. (2014) The potential protective effect of Physalis peruviana L. against carbon tetrachloride-induced hepatotoxicity in rats is mediated by suppression of oxidative stress and downregulation of MMP-9 expression. Oxid Med Cell Longev 2014:381413. - PMC - PubMed
-
- Amacher DE, Adler R, Herath A, Townsend RR. (2005) Use of proteomic methods to identify serum biomarkers associated with rat liver toxicity or hypertrophy. Clin Chem 51:1796–1803. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

