Pharmacological investigation on the anti-oxidant and anti-inflammatory activity of N-acetylcysteine in an ex vivo model of COPD exacerbation
- PMID: 28118826
- PMCID: PMC5260037
- DOI: 10.1186/s12931-016-0500-y
Pharmacological investigation on the anti-oxidant and anti-inflammatory activity of N-acetylcysteine in an ex vivo model of COPD exacerbation
Abstract
Background: Oxidative stress is recognized to be one of predisposing factor in the pathogenesis of COPD. The oxidant/antioxidant imbalance is significantly pronounced in patients with COPD exacerbation. N-acetylcysteine (NAC) seems to be able to reduce COPD exacerbations by modulating the oxidative stress in addition to its well-known mucolytic activity, but there are discordant findings on the actual anti-oxidant activity of NAC.
Methods: The anti-oxidant effect of NAC and its impact on the inflammatory response have been pharmacologically characterized on a human ex vivo model of COPD exacerbation induced by lipopolysaccharide (LPS).
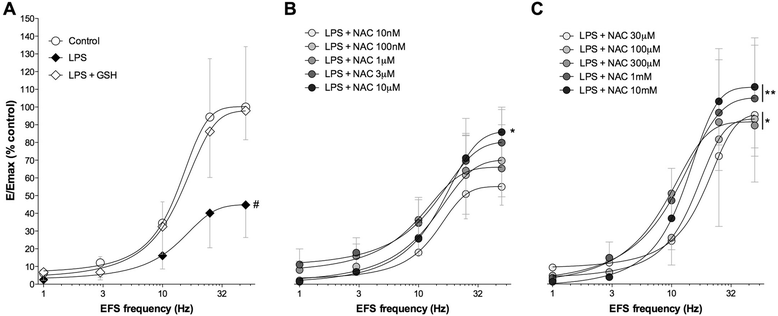
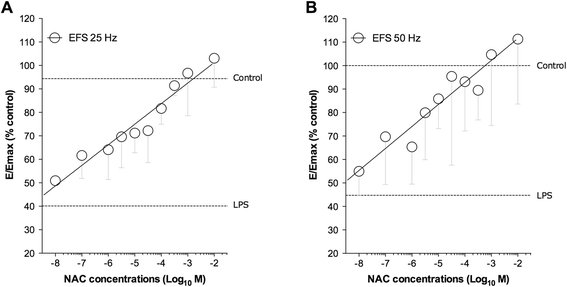
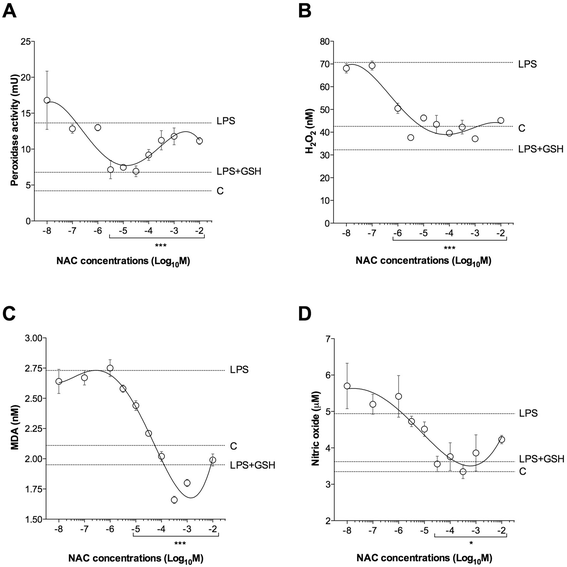
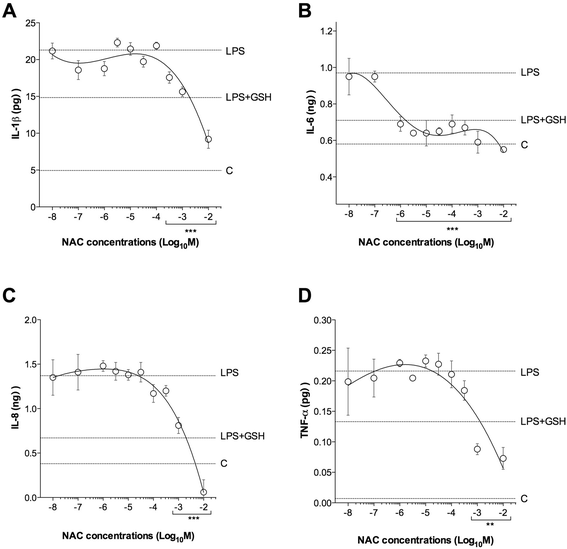
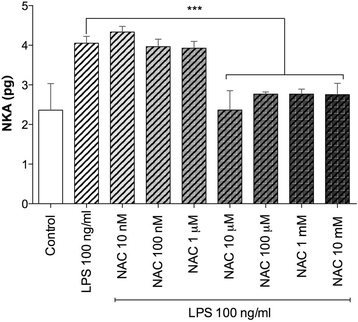
Results: NAC prevented the desensitization induced by LPS incubation on the contractile tone in linear concentration-response manner. Concentrations of NAC ≥1 μM reduced the pro-oxidant response (peroxidase activity, hydrogen peroxide, malondialdehyde, nitric oxide), and improved the anti-oxidant response (total anti-oxidant capacity, glutathione, superoxide dismutase) induced by LPS. Lower concentrations of NAC (<1 μM) did not modulate the bronchial oxidative imbalance. Concentrations of NAC ≥300 μM inhibited the inflammatory response (release of IL-1β, IL-8, and TNF-α) of human airways induced by the overnight stimulation with LPS, whereas lower concentrations of NAC (≥1 μM) were sufficient to reduce the release of IL-6 elicited by LPS. Both the anti-oxidant effect and the anti-inflammatory effect of NAC were inversely correlated with the release of NKA.
Conclusions: The findings of this study suggest that NAC may have a role in modulating the detrimental effect induced by LPS in course of COPD exacerbation. It may elicit both anti-oxidant and anti-inflammatory effects when administered at high concentrations.
Keywords: Anti-inflammatory effect; Anti-oxidant effect; COPD; Lipopolysaccharide; N-acetylcysteine.
Figures

References
-
- Zheng JP, Wen FQ, Bai CX, Wan HY, Kang J, Chen P, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir med. 2014;2:187–94. doi: 10.1016/S2213-2600(13)70286-8. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

