Rationale, design and baseline characteristics of the CANagliflozin cardioVascular Assessment Study-Renal (CANVAS-R): A randomized, placebo-controlled trial
- PMID: 28120497
- PMCID: PMC5348724
- DOI: 10.1111/dom.12829
Rationale, design and baseline characteristics of the CANagliflozin cardioVascular Assessment Study-Renal (CANVAS-R): A randomized, placebo-controlled trial
Abstract
Aims: The primary aim of the CANagliflozin cardioVascular Assessment Study-Renal (CANVAS-R) is to determine whether the favourable effects of inhibition of the sodium glucose co-transporter 2 (SGLT2) on blood glucose, blood pressure and body weight are accompanied by protection against adverse renal outcomes.
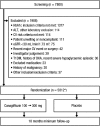
Materials and methods: CANVAS-R is a prospective, randomized, double-blind, placebo-controlled trial in patients with type 2 diabetes with a history or high risk of cardiovascular events. Patients were randomly assigned to once-daily placebo or canagliflozin 100 mg (with optional uptitration to 300 mg) for a planned average of 2.5 years of follow-up. The primary outcome is kidney disease progression, defined by class change in albuminuria. The two secondary outcomes are the composite of hospitalized heart failure or cardiovascular death, and cardiovascular death alone. Effects on end-stage renal disease and a range of other outcomes will also be explored.
Results: A total of 5812 participants were recruited at 422 sites in 24 countries between January 2014 and May 2015. The mean baseline age was 64 years, mean duration of diabetes was 14 years, mean glycated haemoglobin level was 8.3% and mean body mass index was 32 kg/m2 . Of these participants, 37% were women, 71% had a history of cardiovascular disease, 22.3% had microalbuminuria and 8.7% had macroalbuminuria. The mean baseline estimated glomerular filtration rate was 76 mL/min/1.73 m2 . The study will have at least 90% power ( P = .05) to detect a 22% or greater reduction in the risk of progression of albuminuria.
Conclusions: The trial should define the potential renoprotective effect of canagliflozin and will provide additional important new data about its effects on vascular outcomes, death and kidney failure.
Keywords: SGLT2 inhibitor; cardiovascular disease; type 2 diabetes.
© 2017 The Authors. Diabetes, Obesity and Metabolism published by John Wiley & Sons Ltd.
Figures

References
-
- United States Renal Data Systems . USRDS annual data report: epidemiology of kidney disease in the United States. 2015. http://www.usrds.org/2015/view/Default.aspx. Accessed October 6, 2016.
-
- Parving HH, Lehnert H, Brochner‐Mortensen J, Gomis R, Andersen S, Arner P. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med. 2001;345(12):870‐878. - PubMed
-
- Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345(12):861‐869. - PubMed
-
- Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin‐receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345(12):851‐860. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

