Histochemical Examination on Periodontal Tissues of Klotho-Deficient Mice Fed With Phosphate-Insufficient Diet
- PMID: 28122194
- PMCID: PMC5407563
- DOI: 10.1369/0022155416689670
Histochemical Examination on Periodontal Tissues of Klotho-Deficient Mice Fed With Phosphate-Insufficient Diet
Abstract
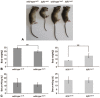
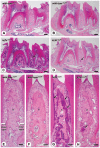
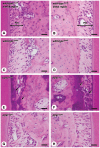
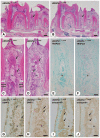
To elucidate which of elevated serum concentration of inorganic phosphate (Pi) or disrupted signaling linked to αklotho/fibroblast growth factor 23 (FGF23) is a predominant regulator for senescence-related degeneration seen in αKlotho-deficient mice, we have examined histological alteration of the periodontal tissues in the mandibular interalveolar septum of αKlotho-deficient mice fed with Pi-insufficient diet. We prepared six groups of mice: wild-type, kl/kl, and αKlotho-/- mice with normal diet or low-Pi diet. As a consequence, kl/klnorPi and αKlotho-/-norPi mice showed the same abnormalities in periodontal tissues: intensely stained areas with hematoxylin in the interalveolar septum, dispersed localization of alkaline phosphatase-positive osteoblasts and tartrate-resistant acid phosphatase-reactive osteoclasts, and accumulation of dentin matrix protein 1 in the osteocytic lacunae. Although kl/kllowPi mice improved these histological abnormalities, αKlotho-/- lowPi mice failed to normalize those. Gene expression of αKlotho was shown to be increased in kl/kl lowPi specimens. It seems likely that histological abnormalities of kl/kl mice have been improved by the rescued expression of αKlotho, rather than low concentration of serum Pi. Thus, the histological malformation in periodontal tissues in αKlotho-deficient mice appears to be due to not only increased concentration of Pi but also disrupted αklotho/FGF23 signaling.
Keywords: DMP-1; osteopontin; periodontal tissue; phosphate; αKlotho-deficient mice.
Conflict of interest statement
Figures




Similar articles
-
The tripartite interaction of phosphate, autophagy, and αKlotho in health maintenance.FASEB J. 2020 Feb;34(2):3129-3150. doi: 10.1096/fj.201902127R. Epub 2020 Jan 5. FASEB J. 2020. PMID: 31908069 Free PMC article.
-
[alpha Klotho and inorganic phosphate metabolism].Clin Calcium. 2007 May;17(5):698-703. Clin Calcium. 2007. PMID: 17470998 Review. Japanese.
-
Altered distribution of bone matrix proteins and defective bone mineralization in klotho-deficient mice.Bone. 2013 Nov;57(1):206-19. doi: 10.1016/j.bone.2013.08.008. Epub 2013 Aug 14. Bone. 2013. PMID: 23954506
-
Correlation between hyperphosphatemia and type II Na-Pi cotransporter activity in klotho mice.Am J Physiol Renal Physiol. 2007 Feb;292(2):F769-79. doi: 10.1152/ajprenal.00248.2006. Epub 2006 Sep 19. Am J Physiol Renal Physiol. 2007. PMID: 16985213
-
Role of αKlotho and FGF23 in regulation of type II Na-dependent phosphate co-transporters.Pflugers Arch. 2019 Jan;471(1):99-108. doi: 10.1007/s00424-018-2238-5. Epub 2018 Dec 1. Pflugers Arch. 2019. PMID: 30506274 Free PMC article. Review.
Cited by
-
Distinct role of Klotho in long bone and craniofacial bone: skeletal development, repair and regeneration.PeerJ. 2024 Oct 21;12:e18269. doi: 10.7717/peerj.18269. eCollection 2024. PeerJ. 2024. PMID: 39465174 Free PMC article. Review.
-
Role of sodium-dependent Pi transporter/Npt2c on Pi homeostasis in klotho knockout mice different properties between juvenile and adult stages.Physiol Rep. 2020 Feb;8(3):e14324. doi: 10.14814/phy2.14324. Physiol Rep. 2020. PMID: 32026654 Free PMC article.
-
Phylogenetic analysis and expression profiling of the Klotho gene family in the short-lived African killifish Nothobranchius furzeri.Dev Genes Evol. 2018 Dec;228(6):255-265. doi: 10.1007/s00427-018-0619-6. Epub 2018 Sep 3. Dev Genes Evol. 2018. PMID: 30178389 Free PMC article.
-
Serum α-Klotho associated with oral health among a nationally representative sample of US adults.Front Endocrinol (Lausanne). 2022 Sep 20;13:970575. doi: 10.3389/fendo.2022.970575. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36204099 Free PMC article.
-
Individual nutrients and serum klotho levels in adults aged 40-79 years.Food Sci Nutr. 2023 Mar 8;11(6):3279-3286. doi: 10.1002/fsn3.3310. eCollection 2023 Jun. Food Sci Nutr. 2023. PMID: 37324910 Free PMC article.
References
-
- Skougaard MR, Frandsen A, Baker DG. Collagen metabolism of skin and periodontal membrane in the squirrel monkey. Scand J Dent Res. 1970;78:374–7. - PubMed
-
- Fullmer HM, Sheetz JH, Narkates AJ. Oxytalan connective tissue fibers: a review. J Oral Pathol. 1974;3:291–316. - PubMed
-
- Rippin JW. Collagen turnover in the periodontal ligament under normal and altered functional forces. I. Young rat molars. J Periodontal Res. 1976;11:101–7. - PubMed
-
- Sodek J. A comparison of the rates of synthesis and turnover of collagen and non-collagen proteins in adult rat periodontal tissues and skin using a microassay. Arch Oral Biol. 1977;22:655–65. - PubMed
-
- Rippin JW. Collagen turnover in the periodontal ligament under normal and altered functional forces. II. Adult rat molars. J Periodontal Res. 1978;13:149–54. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

