The Vps13p-Cdc31p complex is directly required for TGN late endosome transport and TGN homotypic fusion
- PMID: 28122955
- PMCID: PMC5294781
- DOI: 10.1083/jcb.201606078
The Vps13p-Cdc31p complex is directly required for TGN late endosome transport and TGN homotypic fusion
Abstract
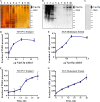
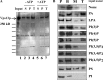
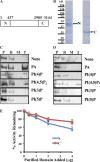
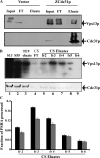
Yeast VPS13 is the founding member of a eukaryotic gene family of growing interest in cell biology and medicine. Mutations in three of four human VPS13 genes cause autosomal recessive neurodegenerative or neurodevelopmental disease, making yeast Vps13p an important structural and functional model. Using cell-free reconstitution with purified Vps13p, we show that Vps13p is directly required both for transport from the trans-Golgi network (TGN) to the late endosome/prevacuolar compartment (PVC) and for TGN homotypic fusion. Vps13p must be in complex with the small calcium-binding protein Cdc31p to be active. Single-particle electron microscopic analysis of negatively stained Vps13p indicates that this 358-kD protein is folded into a compact rod-shaped density (20 × 4 nm) with a loop structure at one end with a circular opening ∼6 nm in diameter. Vps13p exhibits ATP-stimulated binding to yeast membranes and specific interactions with phosphatidic acid and phosphorylated forms of phosphatidyl inositol at least in part through the binding affinities of conserved N- and C-terminal domains.
© 2017 De et al.
Figures


Comment in
-
Vps13 and Cdc31/centrin: Puzzling partners in membrane traffic.J Cell Biol. 2017 Feb;216(2):299-301. doi: 10.1083/jcb.201612026. Epub 2017 Jan 25. J Cell Biol. 2017. PMID: 28122956 Free PMC article.
References
-
- Abazeed M.E., and Fuller R.S.. 2008. Yeast Golgi-localized, gamma-Ear-containing, ADP-ribosylation factor-binding proteins are but adaptor protein-1 is not required for cell-free transport of membrane proteins from the trans-Golgi network to the prevacuolar compartment. Mol. Biol. Cell. 19:4826–4836. 10.1091/mbc.E07-05-0442 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

