Role of TAAR1 within the Subregions of the Mesocorticolimbic Dopaminergic System in Cocaine-Seeking Behavior
- PMID: 28123023
- PMCID: PMC5296782
- DOI: 10.1523/JNEUROSCI.2006-16.2016
Role of TAAR1 within the Subregions of the Mesocorticolimbic Dopaminergic System in Cocaine-Seeking Behavior
Abstract
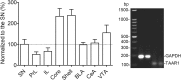
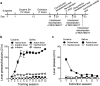
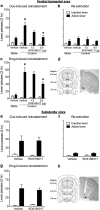
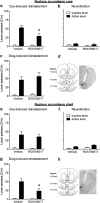
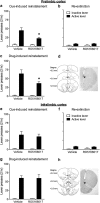
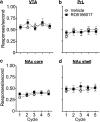
A novel G-protein coupled receptor, trace amine-associated receptor 1 (TAAR1), has been shown to be a promising target to prevent stimulant relapse. Our recent studies showed that systemic administration of TAAR1 agonists decreased abuse-related behaviors of cocaine. However, the role of TAAR1 in specific subregions of the reward system in drug addiction is unknown. Here, using a local pharmacological activation method, we assessed the role of TAAR1 within the subregions of the mesocorticolimbic system: that is, the VTA, the prelimbic cortex (PrL), and infralimbic cortex of medial prefrontal cortex, the core and shell of NAc, BLA, and CeA, on cue- and drug-induced cocaine-seeking in the rat cocaine reinstatement model. We first showed that TAAR1 mRNA was expressed throughout these brain regions. Rats underwent cocaine self-administration, followed by extinction training. RO5166017 (1.5 or 5.0 μg/side) or vehicle was microinjected into each brain region immediately before cue- and drug-induced reinstatement of cocaine-seeking. The results showed that microinjection of RO5166017 into the VTA and PrL decreased both cue- and drug priming-induced cocaine-seeking. Microinjection of RO5166017 into the NAc core and shell inhibited cue- and drug-induced cocaine-seeking, respectively. Locomotor activity or food reinforced operant responding was unaffected by microinjection of RO5166017 into these brain regions. Cocaine-seeking behaviors were not affected by RO5166017 when microinjected into the substantia nigra, infralimbic cortex, BLA, and CeA. Together, these results indicate that TAAR1 in different subregions of the mesocorticolimbic system distinctly contributes to cue- and drug-induced reinstatement of cocaine-seeking behavior.
Significance statement: TAAR1 has been indicated as a modulator of the dopaminergic system. Previous research showed that systemic administration of TAAR1 agonists could attenuate cocaine-related behaviors, suggesting that TAAR1 may be a promising drug target for the treatment of cocaine addiction. However, the specific role of TAAR1 in subregions of the mesocorticolimbic system in drug addiction is unknown. Here, we first showed that TAAR1 mRNA is expressed throughout the subregions of the mesocorticolimbic system. Then, by using a local pharmacological activation method, we demonstrated that TAAR1 in different subregions of the mesocorticolimbic system distinctly contributes to cue- and drug-induced reinstatement of cocaine-seeking behavior.
Keywords: TAAR1; cocaine; mesocorticolimbic regions; reinstatement; self-administration.
Copyright © 2017 the authors 0270-6474/17/370882-11$15.00/0.
Figures

Similar articles
-
Prelimbic to Accumbens Core Pathway Is Recruited in a Dopamine-Dependent Manner to Drive Cued Reinstatement of Cocaine Seeking.J Neurosci. 2016 Aug 17;36(33):8700-11. doi: 10.1523/JNEUROSCI.1291-15.2016. J Neurosci. 2016. PMID: 27535915 Free PMC article.
-
Contribution of a mesocorticolimbic subcircuit to drug context-induced reinstatement of cocaine-seeking behavior in rats.Neuropsychopharmacology. 2014 Feb;39(3):660-9. doi: 10.1038/npp.2013.249. Epub 2013 Sep 20. Neuropsychopharmacology. 2014. PMID: 24051899 Free PMC article.
-
Limbic and motor circuitry underlying footshock-induced reinstatement of cocaine-seeking behavior.J Neurosci. 2004 Feb 18;24(7):1551-60. doi: 10.1523/JNEUROSCI.4177-03.2004. J Neurosci. 2004. PMID: 14973230 Free PMC article.
-
Attenuation of cocaine seeking in rats via enhancement of infralimbic cortical activity using stable step-function opsins.Psychopharmacology (Berl). 2019 Jan;236(1):479-490. doi: 10.1007/s00213-018-4964-y. Epub 2018 Jul 12. Psychopharmacology (Berl). 2019. PMID: 30003306 Free PMC article. Review.
-
Metabotropic glutamate 7 (mGlu7) receptor: a target for medication development for the treatment of cocaine dependence.Neuropharmacology. 2013 Mar;66:12-23. doi: 10.1016/j.neuropharm.2012.04.010. Epub 2012 Apr 21. Neuropharmacology. 2013. PMID: 22546614 Free PMC article. Review.
Cited by
-
Striatal Tyrosine Hydroxylase Is Stimulated via TAAR1 by 3-Iodothyronamine, But Not by Tyramine or β-Phenylethylamine.Front Pharmacol. 2018 Mar 1;9:166. doi: 10.3389/fphar.2018.00166. eCollection 2018. Front Pharmacol. 2018. PMID: 29545750 Free PMC article.
-
TAAR1 agonists attenuate extended-access cocaine self-administration and yohimbine-induced reinstatement of cocaine-seeking.Br J Pharmacol. 2020 Aug;177(15):3403-3414. doi: 10.1111/bph.15061. Epub 2020 May 5. Br J Pharmacol. 2020. PMID: 32246467 Free PMC article.
-
TAAR1 and Psychostimulant Addiction.Cell Mol Neurobiol. 2020 Mar;40(2):229-238. doi: 10.1007/s10571-020-00792-8. Epub 2020 Jan 23. Cell Mol Neurobiol. 2020. PMID: 31974906 Free PMC article. Review.
-
Trace Amine Associated Receptor 1 (TAAR1) Modulation of Food Reward.Front Pharmacol. 2018 Feb 27;9:129. doi: 10.3389/fphar.2018.00129. eCollection 2018. Front Pharmacol. 2018. PMID: 29535626 Free PMC article. Review.
-
Computational Systems Pharmacology-Target Mapping for Fentanyl-Laced Cocaine Overdose.ACS Chem Neurosci. 2019 Aug 21;10(8):3486-3499. doi: 10.1021/acschemneuro.9b00109. Epub 2019 Jul 15. ACS Chem Neurosci. 2019. PMID: 31257858 Free PMC article.
References
-
- Alvarsson A, Zhang X, Stan TL, Schintu N, Kadkhodaei B, Millan MJ, Perlmann T, Svenningsson P. Modulation by trace amine-associated receptor 1 of experimental parkinsonism, l-DOPA responsivity, and glutamatergic neurotransmission. J Neurosci. 2015;35:14057–14069. doi: 10.1523/JNEUROSCI.1312-15.2015. - DOI - PMC - PubMed
-
- Borowsky B, Adham N, Jones KA, Raddatz R, Artymyshyn R, Ogozalek KL, Durkin MM, Lakhlani PP, Bonini JA, Pathirana S, Boyle N, Pu X, Kouranova E, Lichtblau H, Ochoa FY, Branchek TA, Gerald C. Trace amines: identification of a family of mammalian G-protein-coupled receptors. Proc Natl Acad Sci U S A. 2001;98:8966–8971. doi: 10.1073/pnas.151105198. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
