Elevated TMEM106B levels exaggerate lipofuscin accumulation and lysosomal dysfunction in aged mice with progranulin deficiency
- PMID: 28126008
- PMCID: PMC5270347
- DOI: 10.1186/s40478-017-0412-1
Elevated TMEM106B levels exaggerate lipofuscin accumulation and lysosomal dysfunction in aged mice with progranulin deficiency
Abstract
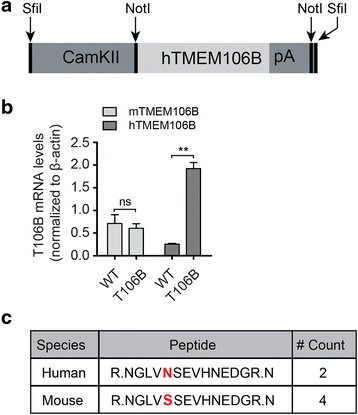
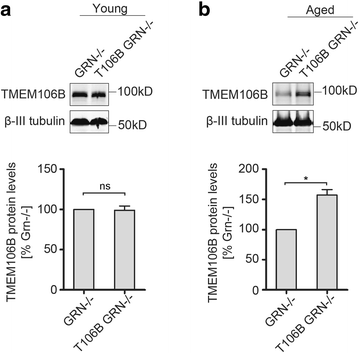
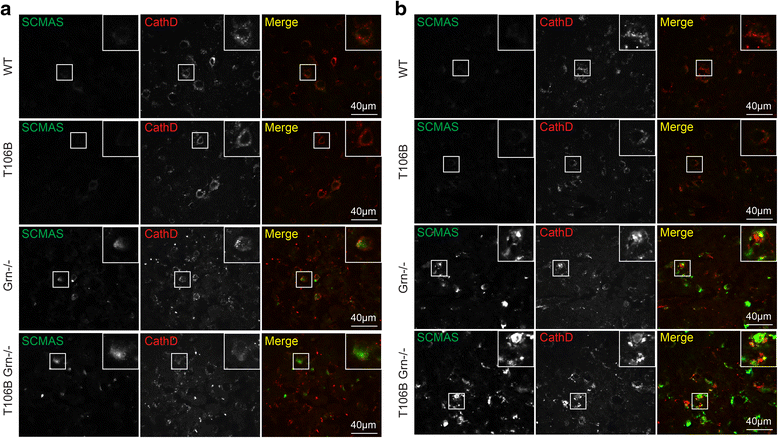
Mutations resulting in haploinsufficiency of progranulin (PGRN) cause frontotemporal lobar degeneration with TDP-43-positive inclusions (FTLD-TDP), a devastating neurodegenerative disease. Accumulating evidence suggest a crucial role of progranulin in maintaining proper lysosomal function during aging. TMEM106B has been identified as a risk factor for frontotemporal lobar degeneration with progranulin mutations and elevated mRNA and protein levels of TMEM106B are associated with increased risk for frontotemporal lobar degeneration. Increased levels of TMEM106B alter lysosomal morphology and interfere with lysosomal degradation. However, how progranulin and TMEM106B interact to regulate lysosomal function and frontotemporal lobar degeneration (FTLD) disease progression is still unclear. Here we report that progranulin deficiency leads to increased TMEM106B protein levels in the mouse cortex with aging. To mimic elevated levels of TMEM106B in frontotemporal lobar degeneration (FTLD) cases, we generated transgenic mice expressing TMEM106B under the neuronal specific promoter, CamKII. Surprisingly, we found that the total protein levels of TMEM106B are not altered despite the expression of the TMEM106B transgene at mRNA and protein levels, suggesting a tight regulation of TMEM106B protein levels in the mouse brain. However, progranulin deficiency results in accumulation of TMEM106B protein from the transgene expression during aging, which is accompanied by exaggerated lysosomal abnormalities and increased lipofuscin accumulation. In summary, our mouse model nicely recapitulates the interaction between progranulin and TMEM106B in human patients and supports a critical role of lysosomal dysfunction in the frontotemporal lobar degeneration (FTLD) disease progression.
Keywords: Frontotemporal lobar degeneration (FTLD); Lipofuscin; Lysosome; Progranulin; TMEM106B.
Figures





References
-
- Ahmed Z, Sheng H, Xu YF, Lin WL, Innes AE, Gass J, Yu X, Wuertzer CA, Hou H, Chiba S, Yamanouchi K, Leissring M, Petrucelli L, Nishihara M, Hutton ML, McGowan E, Dickson DW, Lewis J. Accelerated lipofuscinosis and ubiquitination in granulin knockout mice suggest a role for progranulin in successful aging. Am J Pathol. 2010;177:311–324. doi: 10.2353/ajpath.2010.090915. - DOI - PMC - PubMed
-
- Arai T, Hasegawa M, Akiyama H, Ikeda K, Nonaka T, Mori H, Mann D, Tsuchiya K, Yoshida M, Hashizume Y, Oda T. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun. 2006;351:602–611. doi: 10.1016/j.bbrc.2006.10.093. - DOI - PubMed
-
- Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M. Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature. 2006;442:916–919. doi: 10.1038/nature05016. - DOI - PubMed
-
- Belcastro V, Siciliano V, Gregoretti F, Mithbaokar P, Dharmalingam G, Berlingieri S, Iorio F, Oliva G, Polishchuck R, Brunetti-Pierri N, di Bernardo D. Transcriptional gene network inference from a massive dataset elucidates transcriptome organization and gene function. Nucleic Acids Res. 2011;39:8677–8688. doi: 10.1093/nar/gkr593. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

