Lipidome determinants of maximal lifespan in mammals
- PMID: 28127055
- PMCID: PMC5428381
- DOI: 10.1038/s41598-017-00037-7
Lipidome determinants of maximal lifespan in mammals
Erratum in
-
Publisher Correction: Lipidome determinants of maximal lifespan in mammals.Sci Rep. 2019 May 1;9(1):6972. doi: 10.1038/s41598-019-43122-9. Sci Rep. 2019. PMID: 31043659 Free PMC article.
Abstract
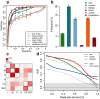
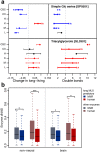
Maximal lifespan of mammalian species, even if closely related, may differ more than 10-fold, however the nature of the mechanisms that determine this variability is unresolved. Here, we assess the relationship between maximal lifespan duration and concentrations of more than 20,000 lipid compounds, measured in 669 tissue samples from 6 tissues of 35 species representing three mammalian clades: primates, rodents and bats. We identify lipids associated with species' longevity across the three clades, uncoupled from other parameters, such as basal metabolic rate, body size, or body temperature. These lipids clustered in specific lipid classes and pathways, and enzymes linked to them display signatures of greater stabilizing selection in long-living species, and cluster in functional groups related to signaling and protein-modification processes. These findings point towards the existence of defined molecular mechanisms underlying variation in maximal lifespan among mammals.
Figures


References
-
- Bonafe M, et al. Polymorphic variants of insulin-like growth factor I (IGF-I) receptor and phosphoinositide 3-kinase genes affect IGF-I plasma levels and human longevity: cues for an evolutionarily conserved mechanism of life span control. The Journal of clinical endocrinology and metabolism. 2003;88:3299–3304. doi: 10.1210/jc.2002-021810. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

