A-FABP mediates adaptive thermogenesis by promoting intracellular activation of thyroid hormones in brown adipocytes
- PMID: 28128199
- PMCID: PMC5290165
- DOI: 10.1038/ncomms14147
A-FABP mediates adaptive thermogenesis by promoting intracellular activation of thyroid hormones in brown adipocytes
Abstract
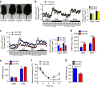
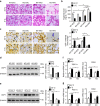
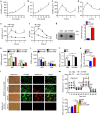
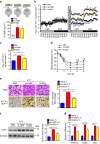
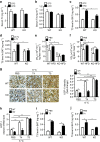
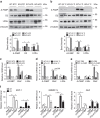
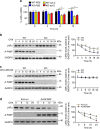
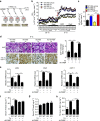
The adipokine adipocyte fatty acid-binding protein (A-FABP) has been implicated in obesity-related cardio-metabolic complications. Here we show that A-FABP increases thermogenesis by promoting the conversion of T4 to T3 in brown adipocytes. We find that A-FABP levels are increased in both white (WAT) and brown (BAT) adipose tissues and the bloodstream in response to thermogenic stimuli. A-FABP knockout mice have reduced thermogenesis and whole-body energy expenditure after cold stress or after feeding a high-fat diet, which can be reversed by infusion of recombinant A-FABP. Mechanistically, A-FABP induces the expression of type-II iodothyronine deiodinase in BAT via inhibition of the nuclear receptor liver X receptor α, thereby leading to the conversion of thyroid hormone from its inactive form T4 to active T3. The thermogenic responses to T4 are abrogated in A-FABP KO mice, but enhanced by A-FABP. Thus, A-FABP acts as a physiological stimulator of BAT-mediated adaptive thermogenesis.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
Similar articles
-
Urolithin A exerts antiobesity effects through enhancing adipose tissue thermogenesis in mice.PLoS Biol. 2020 Mar 27;18(3):e3000688. doi: 10.1371/journal.pbio.3000688. eCollection 2020 Mar. PLoS Biol. 2020. PMID: 32218572 Free PMC article.
-
Adaptive thermogenesis in brown adipose tissue involves activation of pannexin-1 channels.Mol Metab. 2021 Feb;44:101130. doi: 10.1016/j.molmet.2020.101130. Epub 2020 Nov 25. Mol Metab. 2021. PMID: 33248294 Free PMC article.
-
Thyroid hormone (T3) stimulates brown adipose tissue activation via mitochondrial biogenesis and MTOR-mediated mitophagy.Autophagy. 2019 Jan;15(1):131-150. doi: 10.1080/15548627.2018.1511263. Epub 2018 Sep 13. Autophagy. 2019. PMID: 30209975 Free PMC article.
-
Origins and early development of the concept that brown adipose tissue thermogenesis is linked to energy balance and obesity.Biochimie. 2017 Mar;134:62-70. doi: 10.1016/j.biochi.2016.09.007. Epub 2016 Sep 10. Biochimie. 2017. PMID: 27621146 Review.
-
MECHANISMS IN ENDOCRINOLOGY: Brown adipose tissue in humans: regulation and metabolic significance.Eur J Endocrinol. 2016 Jul;175(1):R11-25. doi: 10.1530/EJE-15-1217. Eur J Endocrinol. 2016. PMID: 27220620 Review.
Cited by
-
Intermittent High Glucose Exacerbates A-FABP Activation and Inflammatory Response through TLR4-JNK Signaling in THP-1 Cells.J Immunol Res. 2018 Apr 11;2018:1319272. doi: 10.1155/2018/1319272. eCollection 2018. J Immunol Res. 2018. PMID: 29850615 Free PMC article.
-
Fatty acid binding protein 4 promotes autoimmune diabetes by recruitment and activation of pancreatic islet macrophages.JCI Insight. 2021 Apr 8;6(7):e141814. doi: 10.1172/jci.insight.141814. JCI Insight. 2021. PMID: 33690220 Free PMC article.
-
Transcriptomic profiling of the telomerase transformed Mesenchymal stromal cells derived adipocytes in response to rosiglitazone.BMC Genom Data. 2022 Mar 9;23(1):17. doi: 10.1186/s12863-022-01027-z. BMC Genom Data. 2022. PMID: 35264099 Free PMC article.
-
Associations of three thermogenic adipokines with metabolic syndrome in obese and non-obese populations from the China plateau: the China Multi-Ethnic Cohort.BMJ Open. 2023 Jul 25;13(7):e066789. doi: 10.1136/bmjopen-2022-066789. BMJ Open. 2023. PMID: 37491087 Free PMC article.
-
Adipocyte fatty acid-binding protein exacerbates cerebral ischaemia injury by disrupting the blood-brain barrier.Eur Heart J. 2020 Sep 1;41(33):3169-3180. doi: 10.1093/eurheartj/ehaa207. Eur Heart J. 2020. PMID: 32350521 Free PMC article.
References
-
- Dietrich M. O. & Horvath T. L. Limitations in anti-obesity drug development: the critical role of hunger-promoting neurons. Nat. Rev. Drug Discov. 11, 675–691 (2012). - PubMed
-
- Nedergaard J. et al.. UCP1: the only protein able to mediate adaptive non-shivering thermogenesis and metabolic inefficiency. Biochim. Biophys. Acta 1504, 82–106 (2001). - PubMed
-
- Lowell B. B. & Spiegelman B. M. Towards a molecular understanding of adaptive thermogenesis. Nature 404, 652–660 (2000). - PubMed
-
- Wijers S. L., Saris W. H. & van Marken Lichtenbelt W. D. Recent advances in adaptive thermogenesis: potential implications for the treatment of obesity. Obes. Rev. 10, 218–226 (2009). - PubMed
-
- Cannon B. & Nedergaard J. Nonshivering thermogenesis and its adequate measurement in metabolic studies. J. Exp. Biol. 214, 242–253 (2011). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

