Thrombospondin-1 and microRNA-1 expression in response to multiwalled carbon nanotubes in alveolar epithelial cells
- PMID: 28128526
- PMCID: PMC5392133
- DOI: 10.1002/tox.22387
Thrombospondin-1 and microRNA-1 expression in response to multiwalled carbon nanotubes in alveolar epithelial cells
Abstract
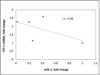
Thrombospondin-1 (TSP-1) is a glycoprotein that plays a role in extracellular matrix (ECM) remodeling. Previously, we have shown that multiwalled carbon nanotubes (MWCNT) regulate ECM components TGFβ and its target Col3A1 in alveolar epithelial cells. In this study, we investigated the effect of MWCNT on TSP-1 and microRNA-1 (miR-1) in the regulation of TGFβ in ECM remodeling using alveolar epithelial A549 cells. A549 cells were treated with MWCNT (20 or 50 µg/mL) for 6 or 24 h and the expression of TSP-1 and miR-1, and the exogenous miR-1 effect on cell morphology were analyzed. MWCNT induced in a time- and dose-dependent manner the expression of TSP-1. miR-1 was suppressed by MWCNT after 6 or 24 h of treatment regardless of the dose. TSP-1 and miR-1 negatively correlated with each other, r = -0.58. Exogenous administration of miR-1 induced alveolar epithelial cell morphology changes including cell clustering, whereas inhibition of miR-1 induced less cell to cell contact, cell rounding, and cellular projections. IntAct molecular network interactions analysis revealed that TSP-1 interacts with 21 molecular factors including ECM genes, and molecules. These results indicate a relationship between that TSP-1, MWCNT, and TGFβ, and suggest TSP-1 may play a role in MWCNT-induced TGFβ and ECM remodeling. Moreover, these data also suggest an inverse relationship between TSP-1 and miR-1 and a potential role of miR-1 in MWCNT-induced fibrotic signaling. © 2016 Wiley Periodicals, Inc. Environ Toxicol 32: 1596-1606, 2017.
Keywords: Alveolar epithelial cells; ECM; MWCNT; Pulmonary fibrosis; TSP-1; miR-1.
© 2017 Wiley Periodicals, Inc.
Figures










Similar articles
-
Effects of lipopolysaccharide, multiwalled carbon nantoubes, and the combination on lung alveolar epithelial cells.Environ Toxicol. 2017 Feb;32(2):445-455. doi: 10.1002/tox.22248. Epub 2016 Feb 16. Environ Toxicol. 2017. PMID: 26880698 Free PMC article.
-
Functional effects of differentially expressed microRNAs in A549 cells exposed to MWCNT-7 or crocidolite.Toxicol Lett. 2020 Aug 1;328:7-18. doi: 10.1016/j.toxlet.2020.04.002. Epub 2020 Apr 18. Toxicol Lett. 2020. PMID: 32311379
-
Thrombospondin-1 mediates multi-walled carbon nanotube induced impairment of arteriolar dilation.Nanotoxicology. 2017 Feb;11(1):112-122. doi: 10.1080/17435390.2016.1277275. Epub 2017 Jan 11. Nanotoxicology. 2017. PMID: 28024456 Free PMC article.
-
Thrombospondin-4 in tissue remodeling.Matrix Biol. 2019 Jan;75-76:300-313. doi: 10.1016/j.matbio.2017.11.006. Epub 2017 Nov 11. Matrix Biol. 2019. PMID: 29138119 Free PMC article. Review.
-
Thrombospondin-1: A Key Protein That Induces Fibrosis in Diabetic Complications.J Diabetes Res. 2020 Jun 11;2020:8043135. doi: 10.1155/2020/8043135. eCollection 2020. J Diabetes Res. 2020. PMID: 32626782 Free PMC article. Review.
Cited by
-
Thrombospondin-1 Airway Expression and Thrombospondin-1 Gene Variants Are Associated with Bronchopulmonary Dysplasia in Extremely Low-Birth-Weight Infants: A Pilot Study.Children (Basel). 2025 Mar 28;12(4):424. doi: 10.3390/children12040424. Children (Basel). 2025. PMID: 40338233 Free PMC article.
-
MIP-4 is Induced by Bleomycin and Stimulates Cell Migration Partially via Nir-1 Receptor.Biochem Res Int. 2024 Aug 2;2024:5527895. doi: 10.1155/2024/5527895. eCollection 2024. Biochem Res Int. 2024. PMID: 39132322 Free PMC article.
-
Idiopathic Pulmonary Comorbidities and Mechanisms.Int J Inflam. 2021 Oct 13;2021:3963659. doi: 10.1155/2021/3963659. eCollection 2021. Int J Inflam. 2021. PMID: 34691383 Free PMC article. Review.
-
miR221 regulates cell migration by targeting annexin a1 expression in human mesothelial MeT-5A cells neoplastic-like transformed by multi-walled carbon nanotube.Genes Environ. 2021 Aug 2;43(1):34. doi: 10.1186/s41021-021-00209-y. Genes Environ. 2021. PMID: 34340715 Free PMC article.
References
-
- Adams JC, Tucker RP, Lawler J. The thrombospondin gene family. Austin: R G Landis; 1995. p. 191.
-
- Albo D, Shinohara T, Tuszynski GP. Up-regulation of matrix metalloproteinase 9 by thrombospondin 1 in gastric cancer. J Surgical Res. 2002;108:51–60. - PubMed
-
- Aragon M, Erdely A, Bishop I, Salmen R, Weaver J, Liu J, Hall P, Eye T, Kodali V, Zeidler-Erdely P, Stafflinger JE, Ottens AK, Campen MJ. MMP-9-dependent serum-borne bioactivity caused by multiwalled carbon nanotubes exposure induces vascular dysfunction via the CD36 scavenger receptor. Toxicol Sci. 2016;150:488–498. - PMC - PubMed
-
- Azuma A, Li Y, Abe S. Interferon-β inhibits bleomycin-induced lung fibrosis by decreasing transforming growth factor-β and thrombospondin. Am J Resp Cell Mol Biol. 2005;32:93–98. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

