Allosteric "beta-blocker" isolated from a DNA-encoded small molecule library
- PMID: 28130548
- PMCID: PMC5321036
- DOI: 10.1073/pnas.1620645114
Allosteric "beta-blocker" isolated from a DNA-encoded small molecule library
Abstract
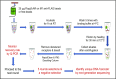
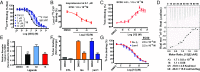
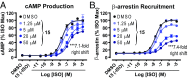
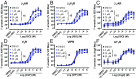
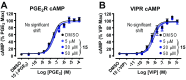
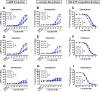
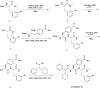
The β2-adrenergic receptor (β2AR) has been a model system for understanding regulatory mechanisms of G-protein-coupled receptor (GPCR) actions and plays a significant role in cardiovascular and pulmonary diseases. Because all known β-adrenergic receptor drugs target the orthosteric binding site of the receptor, we set out to isolate allosteric ligands for this receptor by panning DNA-encoded small-molecule libraries comprising 190 million distinct compounds against purified human β2AR. Here, we report the discovery of a small-molecule negative allosteric modulator (antagonist), compound 15 [([4-((2S)-3-(((S)-3-(3-bromophenyl)-1-(methylamino)-1-oxopropan-2-yl)amino)-2-(2-cyclohexyl-2-phenylacetamido)-3-oxopropyl)benzamide], exhibiting a unique chemotype and low micromolar affinity for the β2AR. Binding of 15 to the receptor cooperatively enhances orthosteric inverse agonist binding while negatively modulating binding of orthosteric agonists. Studies with a specific antibody that binds to an intracellular region of the β2AR suggest that 15 binds in proximity to the G-protein binding site on the cytosolic surface of the β2AR. In cell-signaling studies, 15 inhibits cAMP production through the β2AR, but not that mediated by other Gs-coupled receptors. Compound 15 also similarly inhibits β-arrestin recruitment to the activated β2AR. This study presents an allosteric small-molecule ligand for the β2AR and introduces a broadly applicable method for screening DNA-encoded small-molecule libraries against purified GPCR targets. Importantly, such an approach could facilitate the discovery of GPCR drugs with tailored allosteric effects.
Keywords: DNA-encoded small-molecule library; G-protein–coupled receptor; allosteric modulator; drug discovery; β2-adrenergic receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

