Thermodynamics of manganese oxides: Sodium, potassium, and calcium birnessite and cryptomelane
- PMID: 28130549
- PMCID: PMC5320975
- DOI: 10.1073/pnas.1620427114
Thermodynamics of manganese oxides: Sodium, potassium, and calcium birnessite and cryptomelane
Abstract
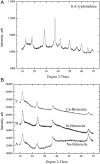
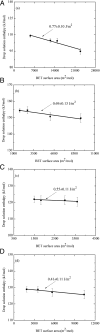
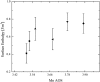
Manganese oxides with layer and tunnel structures occur widely in nature and inspire technological applications. Having variable compositions, these structures often are found as small particles (nanophases). This study explores, using experimental thermochemistry, the role of composition, oxidation state, structure, and surface energy in the their thermodynamic stability. The measured surface energies of cryptomelane, sodium birnessite, potassium birnessite and calcium birnessite are all significantly lower than those of binary manganese oxides (Mn3O4, Mn2O3, and MnO2), consistent with added stabilization of the layer and tunnel structures at the nanoscale. Surface energies generally decrease with decreasing average manganese oxidation state. A stabilizing enthalpy contribution arises from increasing counter-cation content. The formation of cryptomelane from birnessite in contact with aqueous solution is favored by the removal of ions from the layered phase. At large surface area, surface-energy differences make cryptomelane formation thermodynamically less favorable than birnessite formation. In contrast, at small to moderate surface areas, bulk thermodynamics and the energetics of the aqueous phase drive cryptomelane formation from birnessite, perhaps aided by oxidation-state differences. Transformation among birnessite phases of increasing surface area favors compositions with lower surface energy. These quantitative thermodynamic findings explain and support qualitative observations of phase-transformation patterns gathered from natural and synthetic manganese oxides.
Keywords: birnessite; calorimetry; cryptomelane; manganese oxides; thermodynamics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Diversity in the Family of Manganese Oxides at the Nanoscale: From Fundamentals to Applications.ACS Omega. 2020 Oct 5;5(40):25493-25504. doi: 10.1021/acsomega.0c03455. eCollection 2020 Oct 13. ACS Omega. 2020. PMID: 33073076 Free PMC article. Review.
-
Cd2+ adsorption performance of tunnel-structured manganese oxides driven by electrochemically controlled redox.Environ Pollut. 2019 Jan;244:783-791. doi: 10.1016/j.envpol.2018.10.062. Epub 2018 Oct 24. Environ Pollut. 2019. PMID: 30388682
-
[Investigation on the oxidation behaviors and kinetics of sulfide by cryptomelane].Huan Jing Ke Xue. 2011 Jul;32(7):2102-8. Huan Jing Ke Xue. 2011. PMID: 21922837 Chinese.
-
Organic matter interactions with natural manganese oxide and synthetic birnessite.Sci Total Environ. 2017 Apr 1;583:487-495. doi: 10.1016/j.scitotenv.2017.01.120. Epub 2017 Jan 23. Sci Total Environ. 2017. PMID: 28126279
-
A review on the transformation of birnessite in the environment: Implication for the stabilization of heavy metals.J Environ Sci (China). 2024 May;139:496-515. doi: 10.1016/j.jes.2023.06.019. Epub 2023 Jun 20. J Environ Sci (China). 2024. PMID: 38105072 Review.
Cited by
-
Nanoscale Hydration in Layered Manganese Oxides.Langmuir. 2021 Jan 19;37(2):666-674. doi: 10.1021/acs.langmuir.0c02592. Epub 2021 Jan 6. Langmuir. 2021. PMID: 33404244 Free PMC article.
-
A Hollow-Structured Manganese Oxide Cathode for Stable Zn-MnO₂ Batteries.Nanomaterials (Basel). 2018 May 5;8(5):301. doi: 10.3390/nano8050301. Nanomaterials (Basel). 2018. PMID: 29734746 Free PMC article.
-
Understanding the role of manganese oxides in retaining harmful metals: Insights into oxidation and adsorption mechanisms at microstructure level.Eco Environ Health. 2024 Jan 23;3(1):89-106. doi: 10.1016/j.eehl.2024.01.002. eCollection 2024 Mar. Eco Environ Health. 2024. PMID: 38445215 Free PMC article. Review.
-
Diversity in the Family of Manganese Oxides at the Nanoscale: From Fundamentals to Applications.ACS Omega. 2020 Oct 5;5(40):25493-25504. doi: 10.1021/acsomega.0c03455. eCollection 2020 Oct 13. ACS Omega. 2020. PMID: 33073076 Free PMC article. Review.
-
Synthesis, Characterization, and Optimization of Magnetoelectric BaTiO3-Iron Oxide Core-Shell Nanoparticles.Nanomaterials (Basel). 2020 Mar 20;10(3):563. doi: 10.3390/nano10030563. Nanomaterials (Basel). 2020. PMID: 32245105 Free PMC article.
References
-
- Brown GE, Jr, Calas G. Mineral-aqueous solution interfaces and their impact on the environment. Geochem Perspect. 2013;1(4,5):483–742.
-
- Brock SL, et al. A review of porous manganese oxide materials. Chem Mater. 1998;10(10):2619–2628.
-
- Drits VA, Silvester E, Gorshkov AI, Manceau A. Structure of synthetic monoclinic Na-rich birnessite and hexagonal birnessite: I. Results from X-ray diffraction and selected-area electron diffraction. Am Mineral. 1997;82:946–961.
-
- Tebo BM, et al. Biogenic manganese oxides: Properties and mechanisms of formation. Annu Rev Earth Planet Sci. 2004;32:287–328.
-
- Tebo BM, Geszvain K, Lee SW. Geomicrobiology: Molecular and environmental perspective. In: Barton BL, Mandl M, Loy A, editors. The Molecular Geomicrobiology of Bacterial Manganese(II) Oxidation. Springer; Dordrecht, The Netherlands: 2010. pp. 285–308.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

