Autophagy facilitates macrophage depots of sustained-release nanoformulated antiretroviral drugs
- PMID: 28134625
- PMCID: PMC5330738
- DOI: 10.1172/JCI90025
Autophagy facilitates macrophage depots of sustained-release nanoformulated antiretroviral drugs
Abstract
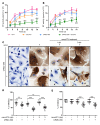
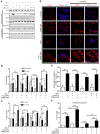
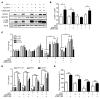
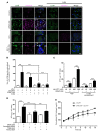
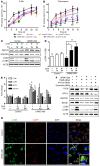
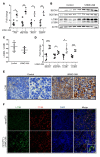
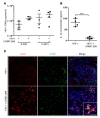
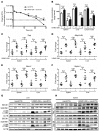
Long-acting anti-HIV products can substantively change the standard of care for patients with HIV/AIDS. To this end, hydrophobic antiretroviral drugs (ARVs) were recently developed for parenteral administration at monthly or longer intervals. While shorter-acting hydrophilic drugs can be made into nanocarrier-encased prodrugs, the nanocarrier encasement must be boosted to establish long-acting ARV depots. The mixed-lineage kinase 3 (MLK-3) inhibitor URMC-099 provides this function by affecting autophagy. Here, we have shown that URMC-099 facilitates ARV sequestration and its antiretroviral responses by promoting the nuclear translocation of the transcription factor EB (TFEB). In monocyte-derived macrophages, URMC-099 induction of autophagy led to retention of nanoparticles containing the antiretroviral protease inhibitor atazanavir. These nanoparticles were localized within macrophage autophagosomes, leading to a 4-fold enhancement of mitochondrial and cell vitality. In rodents, URMC-099 activation of autophagy led to 50-fold increases in the plasma drug concentration of the viral integrase inhibitor dolutegravir. These data paralleled URMC-099-mediated induction of autophagy and the previously reported antiretroviral responses in HIV-1-infected humanized mice. We conclude that pharmacologic induction of autophagy provides a means to extend the action of a long-acting, slow, effective release of antiretroviral therapy.
Conflict of interest statement
Figures
Similar articles
-
Modulating cellular autophagy for controlled antiretroviral drug release.Nanomedicine (Lond). 2018 Sep;13(17):2139-2154. doi: 10.2217/nnm-2018-0224. Epub 2018 Aug 21. Nanomedicine (Lond). 2018. PMID: 30129397 Free PMC article.
-
The mixed lineage kinase-3 inhibitor URMC-099 improves therapeutic outcomes for long-acting antiretroviral therapy.Nanomedicine. 2016 Jan;12(1):109-22. doi: 10.1016/j.nano.2015.09.009. Epub 2015 Oct 22. Nanomedicine. 2016. PMID: 26472049 Free PMC article.
-
Endosomal trafficking of nanoformulated antiretroviral therapy facilitates drug particle carriage and HIV clearance.J Virol. 2014 Sep 1;88(17):9504-13. doi: 10.1128/JVI.01557-14. Epub 2014 Jun 11. J Virol. 2014. PMID: 24920821 Free PMC article.
-
HIV-1 integrase strand transfer inhibitors: a review of current drugs, recent advances and drug resistance.Int J Antimicrob Agents. 2021 May;57(5):106343. doi: 10.1016/j.ijantimicag.2021.106343. Epub 2021 Apr 11. Int J Antimicrob Agents. 2021. PMID: 33852932 Review.
-
Perspectives on the Barrier to Resistance for Dolutegravir + Lamivudine, a Two-Drug Antiretroviral Therapy for HIV-1 Infection.AIDS Res Hum Retroviruses. 2020 Jan;36(1):13-18. doi: 10.1089/AID.2019.0171. Epub 2019 Oct 21. AIDS Res Hum Retroviruses. 2020. PMID: 31507204 Free PMC article. Review.
Cited by
-
LM11A-31, a modulator of p75 neurotrophin receptor, suppresses HIV-1 replication and inflammatory response in macrophages.Exp Biol Med (Maywood). 2024 Jul 25;249:10123. doi: 10.3389/ebm.2024.10123. eCollection 2024. Exp Biol Med (Maywood). 2024. PMID: 39119118 Free PMC article.
-
Creation of a long-acting nanoformulated dolutegravir.Nat Commun. 2018 Feb 6;9(1):443. doi: 10.1038/s41467-018-02885-x. Nat Commun. 2018. PMID: 29402886 Free PMC article.
-
Interplay between Autophagy, Exosomes and HIV-1 Associated Neurological Disorders: New Insights for Diagnosis and Therapeutic Applications.Viruses. 2017 Jul 6;9(7):176. doi: 10.3390/v9070176. Viruses. 2017. PMID: 28684681 Free PMC article. Review.
-
Antiretroviral Drugs Impact Autophagy with Toxic Outcomes.Cells. 2021 Apr 15;10(4):909. doi: 10.3390/cells10040909. Cells. 2021. PMID: 33920955 Free PMC article. Review.
-
A long acting nanoformulated lamivudine ProTide.Biomaterials. 2019 Dec;223:119476. doi: 10.1016/j.biomaterials.2019.119476. Epub 2019 Sep 5. Biomaterials. 2019. PMID: 31525692 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

