The walking dead: macrophage inflammation and death in atherosclerosis
- PMID: 28134664
- PMCID: PMC5808435
- DOI: 10.1097/MOL.0000000000000394
The walking dead: macrophage inflammation and death in atherosclerosis
Abstract
Purpose of review: To highlight recent studies that describe novel inflammatory and signaling mechanisms that regulate macrophage death in atherosclerosis.
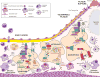
Recent findings: Macrophages contribute to all stages of atherosclerosis. The traditional dogma states that in homeostatic conditions, macrophages undergo apoptosis and are efficiently phagocytosed to be cleared by a process called efferocytosis. In advanced atherosclerosis, however, defective efferocytosis results in secondary necrosis of these uncleared apoptotic cells, which ultimately contributes to the formation of the characteristic necrotic core and the vulnerable plaque. Here, we outline the different types of lesional macrophage death: apoptosis, autophagic and the newly defined necroptosis (i.e. a type of programmed necrosis). Recent discoveries demonstrate that macrophage necroptosis directly contributes to necrotic core formation and plaque instability. Further, promoting the resolution of inflammation using preresolving mediators has been shown to enhance efferocytosis and decrease plaque vulnerability. Finally, the canonical 'don't eat me' signal CD47 has recently been described as playing an important role in atherosclerotic lesion progression by impairing efficient efferocytosis. Although we have made significant strides in improving our understanding of cell death and clearance mechanisms in atherosclerosis, there still remains unanswered questions as to how these pathways can be harnessed using therapeutics to promote lesion regression and disease stability.
Summary: Improving our understanding of the mechanisms that regulate macrophage death in atherosclerosis, in particular apoptosis, necroptosis and efferocytosis, will provide novel therapeutic opportunities to resolve atherosclerosis and promote plaque stability.
Conflict of interest statement
The authors declare that no conflict of interest exists.
Figures
Similar articles
-
Dead cell and debris clearance in the atherosclerotic plaque: Mechanisms and therapeutic opportunities to promote inflammation resolution.Pharmacol Res. 2021 Aug;170:105699. doi: 10.1016/j.phrs.2021.105699. Epub 2021 Jun 2. Pharmacol Res. 2021. PMID: 34087352 Review.
-
Efferocytosis in atherosclerotic lesions: Malfunctioning regulatory pathways and control mechanisms.Pharmacol Ther. 2018 Aug;188:12-25. doi: 10.1016/j.pharmthera.2018.02.003. Epub 2018 Feb 11. Pharmacol Ther. 2018. PMID: 29444453 Review.
-
Macrophage Apoptosis and Necrotic Core Development in Atherosclerosis: A Rapidly Advancing Field with Clinical Relevance to Imaging and Therapy.Can J Cardiol. 2017 Mar;33(3):303-312. doi: 10.1016/j.cjca.2016.12.010. Epub 2016 Dec 21. Can J Cardiol. 2017. PMID: 28232016 Review.
-
The Role of Efferocytosis in Atherosclerosis.Circulation. 2017 Jan 31;135(5):476-489. doi: 10.1161/CIRCULATIONAHA.116.025684. Circulation. 2017. PMID: 28137963 Free PMC article.
-
Macrophage SR-BI mediates efferocytosis via Src/PI3K/Rac1 signaling and reduces atherosclerotic lesion necrosis.J Lipid Res. 2015 Aug;56(8):1449-60. doi: 10.1194/jlr.M056689. Epub 2015 Jun 9. J Lipid Res. 2015. PMID: 26059978 Free PMC article.
Cited by
-
The Role of Immune Checkpoint Molecules on Macrophages in Cancer, Infection, and Autoimmune Pathologies.Front Immunol. 2022 Mar 28;13:837645. doi: 10.3389/fimmu.2022.837645. eCollection 2022. Front Immunol. 2022. PMID: 35418973 Free PMC article. Review.
-
Insights From Pre-Clinical and Clinical Studies on the Role of Innate Inflammation in Atherosclerosis Regression.Front Cardiovasc Med. 2018 May 11;5:32. doi: 10.3389/fcvm.2018.00032. eCollection 2018. Front Cardiovasc Med. 2018. PMID: 29868610 Free PMC article.
-
Oncostatin M promotes the ox-LDL-induced activation of NLRP3 inflammasomes via the NF-κB pathway in THP-1 macrophages and promotes the progression of atherosclerosis.Ann Transl Med. 2022 Apr;10(8):456. doi: 10.21037/atm-22-560. Ann Transl Med. 2022. PMID: 35571419 Free PMC article.
-
Matrine Suppresses Reactive Oxygen Species (ROS)-Mediated MKKs/p38-Induced Inflammation in Oxidized Low-Density Lipoprotein (ox-LDL)-Stimulated Macrophages.Med Sci Monit. 2019 Jun 3;25:4130-4136. doi: 10.12659/MSM.917151. Med Sci Monit. 2019. PMID: 31156213 Free PMC article.
-
Atherosclerosis: The Involvement of Immunity, Cytokines and Cells in Pathogenesis, and Potential Novel Therapeutics.Aging Dis. 2023 Aug 1;14(4):1214-1242. doi: 10.14336/AD.2022.1208. Aging Dis. 2023. PMID: 37163428 Free PMC article. Review.
References
-
- Sakakura K, Nakano M, Otsuka F, Ladich E, Kolodgie FD, Virmani R. Pathophysiology of atherosclerosis plaque progression. Heart Lung Circ. 2013;22:399–411. - PubMed
-
- Yahagi K, Kolodgie FD, Otsuka F, Finn AV, Davis HR, Joner M, et al. Pathophysiology of native coronary, vein graft, and in-stent atherosclerosis. Nat Rev Cardiol. 2016;13:79–98. A comprehensive review of observational pathology of various stages of human atherosclerotic diseases. - PubMed
-
- Shankman LS, Gomez D, Cherepanova OA, Salmon M, Alencar GF, Haskins RM, et al. KLF4-dependent phenotypic modulation of smooth muscle cells has a key role in atherosclerotic plaque pathogenesis. Nat. Med. 2015;21:628–637. A key study using lineage tracing technology re-defined our understanding of the cells involved in atherosclerotic lesions: SMCs can transform into macrophage-like cells in mice. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

