Thienopyrimidinone Based Sirtuin-2 (SIRT2)-Selective Inhibitors Bind in the Ligand Induced Selectivity Pocket
- PMID: 28135086
- PMCID: PMC6014686
- DOI: 10.1021/acs.jmedchem.6b01690
Thienopyrimidinone Based Sirtuin-2 (SIRT2)-Selective Inhibitors Bind in the Ligand Induced Selectivity Pocket
Abstract
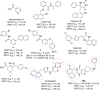
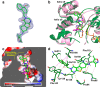
Sirtuins (SIRTs) are NAD-dependent deacylases, known to be involved in a variety of pathophysiological processes and thus remain promising therapeutic targets for further validation. Previously, we reported a novel thienopyrimidinone SIRT2 inhibitor with good potency and excellent selectivity for SIRT2. Herein, we report an extensive SAR study of this chemical series and identify the key pharmacophoric elements and physiochemical properties that underpin the excellent activity observed. New analogues have been identified with submicromolar SIRT2 inhibtory activity and good to excellent SIRT2 subtype-selectivity. Importantly, we report a cocrystal structure of one of our compounds (29c) bound to SIRT2. This reveals our series to induce the formation of a previously reported selectivity pocket but to bind in an inverted fashion to what might be intuitively expected. We believe these findings will contribute significantly to an understanding of the mechanism of action of SIRT2 inhibitors and to the identification of refined, second generation inhibitors.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Selective Sirt2 inhibition by ligand-induced rearrangement of the active site.Nat Commun. 2015 Feb 12;6:6263. doi: 10.1038/ncomms7263. Nat Commun. 2015. PMID: 25672491 Free PMC article.
-
Identification of Diketopiperazine-Containing 2-Anilinobenzamides as Potent Sirtuin 2 (SIRT2)-Selective Inhibitors Targeting the "Selectivity Pocket", Substrate-Binding Site, and NAD+-Binding Site.J Med Chem. 2019 Jun 27;62(12):5844-5862. doi: 10.1021/acs.jmedchem.9b00255. Epub 2019 Jun 12. J Med Chem. 2019. PMID: 31144814
-
Efficient Crystallization of Apo Sirt2 for Small-Molecule Soaking and Structural Analysis of Ligand Interactions.J Med Chem. 2025 Jun 12;68(11):10771-10780. doi: 10.1021/acs.jmedchem.4c02896. Epub 2025 May 20. J Med Chem. 2025. PMID: 40390200 Free PMC article.
-
Selective SIRT2 inhibitors as promising anticancer therapeutics: An update from 2016 to 2020.Eur J Med Chem. 2021 Nov 15;224:113709. doi: 10.1016/j.ejmech.2021.113709. Epub 2021 Jul 18. Eur J Med Chem. 2021. PMID: 34303869 Review.
-
Opening the Selectivity Pocket in the Human Lysine Deacetylase Sirtuin2 - New Opportunities, New Questions.Chem Rec. 2018 Dec;18(12):1701-1707. doi: 10.1002/tcr.201800044. Epub 2018 Jun 21. Chem Rec. 2018. PMID: 29931800 Review.
Cited by
-
The histone deacetylase inhibitor nicotinamide exacerbates neurodegeneration in the lactacystin rat model of Parkinson's disease.J Neurochem. 2019 Jan;148(1):136-156. doi: 10.1111/jnc.14599. Epub 2018 Nov 26. J Neurochem. 2019. PMID: 30269333 Free PMC article.
-
Recent Advances in the Discovery of SIRT1/2 Inhibitors via Computational Methods: A Perspective.Pharmaceuticals (Basel). 2024 May 8;17(5):601. doi: 10.3390/ph17050601. Pharmaceuticals (Basel). 2024. PMID: 38794171 Free PMC article. Review.
-
HaloTag-Targeted Sirtuin-Rearranging Ligand (SirReal) for the Development of Proteolysis-Targeting Chimeras (PROTACs) against the Lysine Deacetylase Sirtuin 2 (Sirt2)*.Chembiochem. 2020 Dec 1;21(23):3371-3376. doi: 10.1002/cbic.202000351. Epub 2020 Aug 27. Chembiochem. 2020. PMID: 32672888 Free PMC article.
-
The crystal structure of the Leishmania infantum Silent Information Regulator 2 related protein 1: Implications to protein function and drug design.PLoS One. 2018 Mar 15;13(3):e0193602. doi: 10.1371/journal.pone.0193602. eCollection 2018. PLoS One. 2018. PMID: 29543820 Free PMC article.
-
Sirtuin activators and inhibitors: Promises, achievements, and challenges.Pharmacol Ther. 2018 Aug;188:140-154. doi: 10.1016/j.pharmthera.2018.03.004. Epub 2018 Mar 22. Pharmacol Ther. 2018. PMID: 29577959 Free PMC article. Review.
References
-
- Du J.; Zhou Y.; Su X.; Yu J. J.; Khan S.; Jiang H.; Kim J.; Woo J.; Kim J. H.; Choi B. H.; He B.; Chen W.; Zhang S.; Cerione R. A.; Auwerx J.; Hao Q.; Lin H. Sirt5 is an NAD-dependent protein lysine demalonylase and desuccinylase. Science 2011, 334, 806–809. 10.1126/science.1207861. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Miscellaneous

