The Metabolism of Methazolamide in Immortalized Human Keratinocytes, HaCaT Cells
- PMID: 28137210
- PMCID: PMC5403967
- DOI: 10.2174/1872312811666170127160931
The Metabolism of Methazolamide in Immortalized Human Keratinocytes, HaCaT Cells
Abstract
Objective: Drug therapy is occasionally accompanied by an idiosyncratic severe toxicity, which occurs very rarely, but can lead to patient mortality. Methazolamide, an anti-glaucomatous agent, could cause severe skin eruptions called Stevens-Johnson syndrome/toxic epidermal necrolyis (SJS/TEN). Its precise etiology is still uncertain. In this study, the metabolism of methazolamide was investigated in immortalized human keratinocytes to reveal the possible mechanism which causes SJS/TEN.
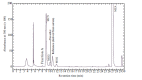
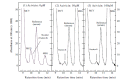
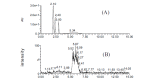
Methods: The metabolism of methazolamide was studied using immortalized human keratinocytes, HaCaT cells. HPLC was used to isolate a metabolite from the culture medium. Mass spectrometry (LCMS/ MS) was employed for its characterization. Three typical chemical inducers were assessed for the inducibility of cytochrome P450, and methimazole was used as the inhibitor of flavin-containing monooxygenase (FMO).
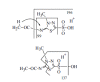
Results: A sulfonic acid, N-[3-methyl-5-sulfo-1,3,4-thiadiazol-2(3H)-ylidene]acetamide (MSO) was identified as the final metabolite. Dexamethasone and β-naphthoflavone behaved as an inducer of cytochrome P450 in the metabolism, but isoniazid did not. The effect of methimazole was not consistent. We did not detect any glucuronide nor any mercapturic acid (N-acetylcysteine conjugate).
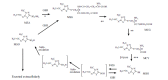
Conclusion: N-[3-methyl-5-sulfo-1,3,4-thiadiazol-2(3H)-ylidene]acetamide (MSO) is not considered to be a direct product of an enzymatic reaction, but rather an auto-oxidation product of N-[3-methyl-5- sulfe-1,3,4-thiadiazol-2(3H)-ylidene]acetamide, a chemically unstable sulfenic acid, which is produced by cytochrome P450 from the β-lyase product of cysteine conjugate of methazolamide. MSO is considered to be susceptible to glutathione and to return to glutathione conjugate of methazolamide, forming a futile cycle. A hypothetical scenario is presented as to the onset of the disease.
Keywords: 3; 4-thiadiazo-2(3H)-ylidene]acetamide; Cytochrome P450; N-[3-methyl-5-sulfo-1; Stevens-Johnson syndrome; glutathione; methazolamide; sulfenic acid; sulfonic acid; toxic epidermal necrolysis.
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.org.
Figures


Similar articles
-
The metabolism of methazolamide - identification of metabolites in guinea pig urine.Drug Metab Lett. 2011 Jan;5(1):45-54. doi: 10.2174/187231211794455299. Drug Metab Lett. 2011. PMID: 21198437
-
Cysteine conjugate of methazolamide is metabolized by beta-lyase.J Pharm Sci. 2001 Feb;90(2):224-33. doi: 10.1002/1520-6017(200102)90:2<224::aid-jps13>3.0.co;2-0. J Pharm Sci. 2001. PMID: 11169539
-
HLA-B*5901 is strongly associated with methazolamide-induced Stevens-Johnson syndrome/toxic epidermal necrolysis.Pharmacogenomics. 2010 Jun;11(6):879-84. doi: 10.2217/pgs.10.54. Pharmacogenomics. 2010. PMID: 20504258
-
[News in severe clinical adverse drug reactions: Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN)].Gac Med Mex. 2015 Nov-Dec;151(6):777-87. Gac Med Mex. 2015. PMID: 26581536 Review. Spanish.
-
Drug interactions with normal and TEN epidermal keratinocytes.Curr Drug Saf. 2012 Nov 1;7(5):352-6. doi: 10.2174/157488612805076507. Curr Drug Saf. 2012. PMID: 23373550 Review.
Cited by
-
In silico, in vitro, and in vivo human metabolism of acetazolamide, a carbonic anhydrase inhibitor and common "diuretic and masking agent" in doping.Arch Toxicol. 2022 Jul;96(7):1989-2001. doi: 10.1007/s00204-022-03289-z. Epub 2022 Apr 11. Arch Toxicol. 2022. PMID: 35410394
References
-
- Hausmann O., Schnyder B., Pichler W.J. Drug hypersensitivity reactions involving skin. In: Uetrecht J., editor. Springer-Verlag. Vol. 196. Berlin, Heidelberg: 2010. pp. 2–55. (Handbook of Experimental Pharmacology). - PubMed
-
- Schwaiblmair M., Behr W., Berghaus W.F. Cytochrome P450 polymorphisms and drug-induced interstitial lung disease. Expert Opin. Drug Metab. Toxicol. 2011;7:1547–1560. - PubMed
-
- Konstantopoulos N., Molero J.C., McGee S.L., Spolding B., Connor T., Vries M., Wanyonyi S., Morrison S., Swinton C., Jones S., Cooper A., Garcia-Guerra L., Foletta V., Krippner G., Andrikopoulos S., Walder K.R. Methazolamide is a new hepatic insulin sensitizer that lowers blood glucose in vivo. Diabetes. 2012;61:2146–2154. - PMC - PubMed
-
- Simpson R.W., Nicolson G.C., Proietto J., Sarah A., Sanders K.M., Phillips G., Chambers J., MacGinley R., Orford N., Walder K., Krippner G., Skoff K., Wacher V.J. Efficacy and safety of oral methazolamide in patients with type 2 diabetes: A 24-week, placebo-contorolled, double-blind study. Diabetes Care. 2014;37:3121–3123. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

