Intramolecular 1,5-C(sp3)-H Radical Amination via Co(II)-Based Metalloradical Catalysis for Five-Membered Cyclic Sulfamides
- PMID: 28138382
- PMCID: PMC5271564
- DOI: 10.1039/C6SC02231F
Intramolecular 1,5-C(sp3)-H Radical Amination via Co(II)-Based Metalloradical Catalysis for Five-Membered Cyclic Sulfamides
Abstract
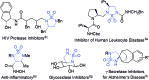
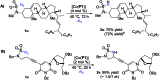
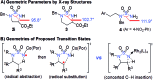
Co(II)-based metalloradical catalysis (MRC) proves effective for intramolecular 1,5-C-H amination of sulfamoyl azides under neutral and nonoxidative conditions, providing a straightforward approach to access strained 5-membered cyclic sulfamides with nitrogen gas as the only byproduct. The metalloradical amination system is applicable to different types of C(sp3)-H bonds and has a high degree of functional group tolerance. Additional features of the Co(II)-catalyzed 1,5-C-H amination include excellent chemoselectivity toward allylic and propargylic C-H bonds. The unique reactivity and selectivity profile of the Co(II)-catalyzed 1,5-C-H amination is attributed to the underlying radical mechanism of MRC.
Figures


Similar articles
-
Catalytic Radical Process for Enantioselective Amination of C(sp3 )-H Bonds.Angew Chem Int Ed Engl. 2018 Dec 17;57(51):16837-16841. doi: 10.1002/anie.201808923. Epub 2018 Nov 16. Angew Chem Int Ed Engl. 2018. PMID: 30347505 Free PMC article.
-
Chemoselective amination of propargylic C(sp³)-H bonds by cobalt(II)-based metalloradical catalysis.Angew Chem Int Ed Engl. 2014 Jul 1;53(27):7028-32. doi: 10.1002/anie.201400557. Epub 2014 May 19. Angew Chem Int Ed Engl. 2014. PMID: 24840605 Free PMC article.
-
Enantioselective Radical Construction of 5-Membered Cyclic Sulfonamides by Metalloradical C-H Amination.J Am Chem Soc. 2019 Nov 13;141(45):18160-18169. doi: 10.1021/jacs.9b08894. Epub 2019 Nov 1. J Am Chem Soc. 2019. PMID: 31622088 Free PMC article.
-
Azides in the Synthesis of Various Heterocycles.Molecules. 2022 Jun 9;27(12):3716. doi: 10.3390/molecules27123716. Molecules. 2022. PMID: 35744839 Free PMC article. Review.
-
Iron-catalysed intramolecular C(sp3)-H amination of alkyl azides.Chem Commun (Camb). 2024 Nov 26;60(95):13998-14011. doi: 10.1039/d4cc04169k. Chem Commun (Camb). 2024. PMID: 39531011 Review.
Cited by
-
Stereocontrolled synthesis of bicyclic ureas and sulfamides via Pd-catalyzed alkene carboamination reactions.Tetrahedron. 2019 Aug 9;75(32):4228-4243. doi: 10.1016/j.tet.2019.04.031. Epub 2019 Apr 20. Tetrahedron. 2019. PMID: 31866698 Free PMC article.
-
Reactions of an Anionic Gallylene with Azobenzene or Azide Compounds Through C(sp2)-H and C(sp3)-H Activation.Molecules. 2024 Oct 24;29(21):5021. doi: 10.3390/molecules29215021. Molecules. 2024. PMID: 39519661 Free PMC article.
-
Metalloradical Catalysis: General Approach for Controlling Reactivity and Selectivity of Homolytic Radical Reactions.Angew Chem Int Ed Engl. 2024 May 13;63(20):e202320243. doi: 10.1002/anie.202320243. Epub 2024 Apr 3. Angew Chem Int Ed Engl. 2024. PMID: 38472114 Free PMC article. Review.
-
Convergent synthesis of thiodiazole dioxides from simple ketones and amines through an unusual nitrogen-migration mechanism.Chem Sci. 2023 Nov 27;15(1):328-335. doi: 10.1039/d3sc04478e. eCollection 2023 Dec 20. Chem Sci. 2023. PMID: 38131085 Free PMC article.
-
Radical cascade synthesis of azoles via tandem hydrogen atom transfer.Chem Sci. 2020 Jan 31;11(9):2479-2486. doi: 10.1039/c9sc06239d. Chem Sci. 2020. PMID: 34084413 Free PMC article.
References
-
-
For selected recent examples, see:
- Du J., Skubi K. L., Schultz D. M., Yoon T. P. Science. 2014;344:392–396. - PMC - PubMed
- Hoyt J. M., Schmidt V. A., Tondreau A. M., Chirik P. J. Science. 2015;349:960–963. - PubMed
- Jeffrey J. L., Terrett J. A., MacMillan D. W. C. Science. 2015;349:1532–1536. - PMC - PubMed
- Kainz Q. M., Matier C. D., Bartoszewicz A., Zultanski S. L., Peters J. C., Fu G. C. Science. 2016;351:681–684. - PMC - PubMed
-
-
-
For reviews on applications of Co(
ii )-based MRC, see: - Fantauzzi S., Caselli A., Gallo E. Dalton Trans. 2009:5434–5443. - PubMed
- Driver T. G. Org. Biomol. Chem. 2010;8:3831–3846. - PMC - PubMed
- Lu H., Zhang X. P. Chem. Soc. Rev. 2011;40:1899–1909. - PubMed
- Che C. M., Lo V. K. Y., Zhou C. Y., Huang J. S. Chem. Soc. Rev. 2011;40:1950–1975. - PubMed
- Pellissier H., Clavier H. Chem. Rev. 2014;114:2775–2823. - PubMed
-
-
-
For a recent review on Ti(
iii )-based MRC, see: . For the first demonstration of Ti(iii )-based metalloradical non-catalytic process, see: . For select examples of catalytic processes via Ti(III)-based MRC, see: - Gansäuer A., Hildebrandt S., Vogelsang E., Flowers II R. A. Dalton Trans. 2016;45:448–452. - PubMed
- Nugent W. A., RajanBabu T. V. J. Am. Chem. Soc. 1988;110:8561–8562.
- Gansäuer A., Rinker B., Pierobon M., Grimme S., Gerenkamp M., Mück-Lichtenfeld C. Angew. Chem., Int. Ed. 2003;42:3687–3690. - PubMed
- Gansäuer A., Fan C.-A., Keller F., Keil J. J. Am. Chem. Soc. 2007;129:3484–3485. - PubMed
- Gansäuer A., Fleckhaus A., Lafont M. A., Okkel A., Kotsis K., Anoop A., Neese F. J. Am. Chem. Soc. 2009;131:16989–16999. - PubMed
- Gansäuer A., Hildebrandt S., Michelmann A., Dahmen T., von Laufenberg D., Kube C., Fianu G. D., Flowers II R. A. Angew. Chem., Int. Ed. 2015;54:7003–7006. - PubMed
-
-
-
For detailed studies on the radical mechanism of [Co(Por)]-catalyzed C–H amination, including EPR observation of α-Co(
iii )-aminyl radical intermediates (also known as Co(iii )-nitrene radicals), see: . For related DFT studies on the radical mechanism of [Co(Por)]-catalyzed olefin aziridination and the ligand hydrogen-bonding effect, see: ; for detailed studies on a similar radical mechanism involving α-Co(iii )-alkyl radical intermediates (also known as Co(iii )-carbene radicals), see: - Lyaskovskyy V., Olivos Suarez A. I., Lu H., Jiang H., Zhang X. P., de Bruin B. J. Am. Chem. Soc. 2011;133:12264–12273. - PubMed
- Olivos Suarez A. I., Jiang H., Zhang X. P., de Bruin B. Dalton Trans. 2011;40:5697–5705. - PubMed
- Hopmann K. H., Ghosh A. ACS Catal. 2011;1:597–600.
- Dzik W. I., Xu X., Zhang X. P., Reek J. N. H., de Bruin B. J. Am. Chem. Soc. 2010;132:10891–10902. - PubMed
- Belof J. L., Cioce C. R., Xu X., Zhang X. P., Space B., Woodcock H. L. Organometallics. 2011;30:2739–2746. - PMC - PubMed
- Lu H., Dzik W. I., Xu X., Wojtas L., de Bruin B., Zhang X. P. J. Am. Chem. Soc. 2011;133:8518–8521. - PubMed
-
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

