Combination treatment using DDX3 and PARP inhibitors induces synthetic lethality in BRCA1-proficient breast cancer
- PMID: 28138868
- PMCID: PMC5576028
- DOI: 10.1007/s12032-017-0889-2
Combination treatment using DDX3 and PARP inhibitors induces synthetic lethality in BRCA1-proficient breast cancer
Abstract
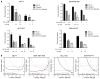
Triple-negative breast cancers have unfavorable outcomes due to their inherent aggressive behavior and lack of targeted therapies. Breast cancers occurring in BRCA1 mutation carriers are mostly triple-negative and harbor homologous recombination deficiency, sensitizing them to inhibition of a second DNA damage repair pathway by, e.g., PARP inhibitors. Unfortunately, resistance against PARP inhibitors in BRCA1-deficient cancers is common and sensitivity is limited in BRCA1-proficient breast cancers. RK-33, an inhibitor of the RNA helicase DDX3, was previously demonstrated to impede non-homologous end-joining repair of DNA breaks. Consequently, we evaluated DDX3 as a therapeutic target in BRCA pro- and deficient breast cancers and assessed whether DDX3 inhibition could sensitize cells to PARP inhibition. High DDX3 expression was identified by immunohistochemistry in breast cancer samples of 24% of BRCA1 (p = 0.337) and 21% of BRCA2 mutation carriers (p = 0.624), as compared to 30% of sporadic breast cancer samples. The sensitivity to the DDX3 inhibitor RK-33 was similar in BRCA1 pro- and deficient breast cancer cell lines, with IC50 values in the low micromolar range (2.8-6.6 μM). A synergistic interaction was observed for combination treatment with RK-33 and the PARP inhibitor olaparib in BRCA1-proficient breast cancer, with the mean combination index ranging from 0.59 to 0.62. Overall, we conclude that BRCA pro- and deficient breast cancers have a similar dependency upon DDX3. DDX3 inhibition by RK-33 synergizes with PARP inhibitor treatment, especially in breast cancers with a BRCA1-proficient background.
Keywords: BRCA; Breast cancer; DDX3; DEAD box RNA helicase; DNA damage repair; PARP inhibitor.
Figures
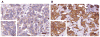
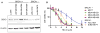
Similar articles
-
Differential Potential of Pharmacological PARP Inhibitors for Inhibiting Cell Proliferation and Inducing Apoptosis in Human Breast Cancer Cells.J Cell Biochem. 2015 Dec;116(12):2824-39. doi: 10.1002/jcb.25229. J Cell Biochem. 2015. PMID: 25981734
-
Co-targeting poly(ADP-ribose) polymerase (PARP) and histone deacetylase (HDAC) in triple-negative breast cancer: Higher synergism in BRCA mutated cells.Biomed Pharmacother. 2018 Mar;99:543-551. doi: 10.1016/j.biopha.2018.01.045. Epub 2018 Feb 20. Biomed Pharmacother. 2018. PMID: 29902865
-
Combined inhibition of PI3K and PARP is effective in the treatment of ovarian cancer cells with wild-type PIK3CA genes.Gynecol Oncol. 2016 Sep;142(3):548-56. doi: 10.1016/j.ygyno.2016.07.092. Epub 2016 Jul 15. Gynecol Oncol. 2016. PMID: 27426307 Free PMC article.
-
Resurrection of PARP Inhibitors in Breast Cancer.J Natl Compr Canc Netw. 2018 Sep;16(9):1150-1156. doi: 10.6004/jnccn.2018.7031. J Natl Compr Canc Netw. 2018. PMID: 30181424 Review.
-
Advances in PARP inhibitors for the treatment of breast cancer.Expert Opin Pharmacother. 2015;16(18):2751-8. doi: 10.1517/14656566.2015.1100168. Epub 2015 Oct 20. Expert Opin Pharmacother. 2015. PMID: 26485111 Review.
Cited by
-
Abnormal expression of YEATS4 associates with poor prognosis and promotes cell proliferation of hepatic carcinoma cell by regulation the TCEA1/DDX3 axis.Am J Cancer Res. 2018 Oct 1;8(10):2076-2087. eCollection 2018. Am J Cancer Res. 2018. PMID: 30416857 Free PMC article.
-
The genomic architecture of EBV and infected gastric tissue from precursor lesions to carcinoma.Genome Med. 2021 Sep 7;13(1):146. doi: 10.1186/s13073-021-00963-2. Genome Med. 2021. PMID: 34493320 Free PMC article.
-
Structural Features of Nucleoprotein CST/Shelterin Complex Involved in the Telomere Maintenance and Its Association with Disease Mutations.Cells. 2020 Feb 4;9(2):359. doi: 10.3390/cells9020359. Cells. 2020. PMID: 32033110 Free PMC article. Review.
-
Functional Roles and Mechanistic Insights of YEATS Domain Proteins in Digestive System Tumors.Dig Dis Sci. 2025 Jun 27. doi: 10.1007/s10620-025-09117-5. Online ahead of print. Dig Dis Sci. 2025. PMID: 40571889 Review.
-
RNA helicase, DDX3X, is actively recruited to sites of DNA damage in live cells.DNA Repair (Amst). 2021 Jul;103:103137. doi: 10.1016/j.dnarep.2021.103137. Epub 2021 May 18. DNA Repair (Amst). 2021. PMID: 34083132 Free PMC article.
References
-
- Moynahan ME, Chiu JW, Koller BH, Jasin M. Brca1 controls homology-directed DNA repair. Mol Cell. 1999;4(4):511–8. - PubMed
-
- Lakhani SR, Van De Vijver MJ, Jacquemier J, Anderson TJ, Osin PP, McGuffog L, et al. The pathology of familial breast cancer: predictive value of immunohistochemical markers estrogen receptor, progesterone receptor, HER-2, and p53 in patients with mutations in BRCA1 and BRCA2. J Clin Oncol. 2002;20(9):2310–8. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

