A radiobiological model of radiotherapy response and its correlation with prognostic imaging variables
- PMID: 28140359
- PMCID: PMC5512557
- DOI: 10.1088/1361-6560/aa5d42
A radiobiological model of radiotherapy response and its correlation with prognostic imaging variables
Abstract
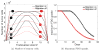
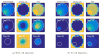
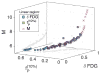
Radiobiological models of tumour control probability (TCP) can be personalized using imaging data. We propose an extension to a voxel-level radiobiological TCP model in order to describe patient-specific differences and intra-tumour heterogeneity. In the proposed model, tumour shrinkage is described by means of a novel kinetic Monte Carlo method for inter-voxel cell migration and tumour deformation. The model captures the spatiotemporal evolution of the tumour at the voxel level, and is designed to take imaging data as input. To test the performance of the model, three image-derived variables found to be predictive of outcome in the literature have been identified and calculated using the model's own parameters. Simulating multiple tumours with different initial conditions makes it possible to perform an in silico study of the correlation of these variables with the dose for 50% tumour control ([Formula: see text]) calculated by the model. We find that the three simulated variables correlate with the calculated [Formula: see text]. In addition, we find that different variables have different levels of sensitivity to the spatial distribution of hypoxia within the tumour, as well as to the dynamics of the migration mechanism. Finally, based on our results, we observe that an adequate combination of the variables may potentially result in higher predictive power.
Figures





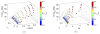
References
-
- Antipas VP, Stamatakos GS, Uzunoglu NK, Dionysiou DD, Dale RG. A spatio-temporal simulation model of the response of solid tumours to radiotherapy in vivo: parametric validation concerning oxygen enhancement ratio and cell cycle duration. Physics in medicine and biology. 2004;49(8):1485–504. - PubMed
-
- van Baardwijk A, et al. Time trends in the maximal uptake of FDG on PET scan during thoracic radiotherapy. A prospective study in locally advanced non-small cell lung cancer (NSCLC) patients. Radiotherapy and Oncology. 2007;82(2):145–152. - PubMed
-
- Baert AL, Hermans R. Head and neck cancer imaging. Springer Science & Business Media; 2006.
-
- Bortz A, Kalos M, Lebowitz J. A new algorithm for Monte Carlo simulation of Ising spin systems. Journal of Computational Physics. 1975;17(1):10–18.
-
- Brenner DJ, Hlatky LR, Hahnfeldt PJ, Huang Y, Sachs RK. The linear-quadratic model and most other common radiobiological models result in similar predictions of time-dose relationships. Radiation research. 1998;150(1):83–91. arXiv: NIHMS150003. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
