Glomerular Hyperfiltration in Diabetes: Mechanisms, Clinical Significance, and Treatment
- PMID: 28143897
- PMCID: PMC5373460
- DOI: 10.1681/ASN.2016060666
Glomerular Hyperfiltration in Diabetes: Mechanisms, Clinical Significance, and Treatment
Abstract
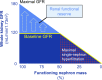
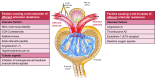
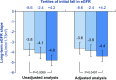
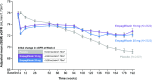
An absolute, supraphysiologic elevation in GFR is observed early in the natural history in 10%-67% and 6%-73% of patients with type 1 and type 2 diabetes, respectively. Moreover, at the single-nephron level, diabetes-related renal hemodynamic alterations-as an adaptation to reduction in functional nephron mass and/or in response to prevailing metabolic and (neuro)hormonal stimuli-increase glomerular hydraulic pressure and transcapillary convective flux of ultrafiltrate and macromolecules. This phenomenon, known as glomerular hyperfiltration, classically has been hypothesized to predispose to irreversible nephron damage, thereby contributing to initiation and progression of kidney disease in diabetes. However, dedicated studies with appropriate diagnostic measures and clinically relevant end points are warranted to confirm this assumption. In this review, we summarize the hitherto proposed mechanisms involved in diabetic hyperfiltration, focusing on ultrastructural, vascular, and tubular factors. Furthermore, we review available evidence on the clinical significance of hyperfiltration in diabetes and discuss currently available and emerging interventions that may attenuate this renal hemodynamic abnormality. The revived interest in glomerular hyperfiltration as a prognostic and pathophysiologic factor in diabetes may lead to improved and timely detection of (progressive) kidney disease, and could provide new therapeutic opportunities in alleviating the renal burden in this population.
Keywords: albuminuria; diabetes; diabetic nephropathy; glomerular filtration rate; glomerular hyperfiltration.
Copyright © 2017 by the American Society of Nephrology.
Figures

References
-
- Tuttle KR, Bakris GL, Bilous RW, Chiang JL, de Boer IH, Goldstein-Fuchs J, Hirsch IB, Kalantar-Zadeh K, Narva AS, Navaneethan SD, Neumiller JJ, Patel UD, Ratner RE, Whaley-Connell AT, Molitch ME: Diabetic kidney disease: A report from an ADA Consensus Conference. Diabetes Care 37: 2864–2883, 2014 - PMC - PubMed
-
- National Kidney Foundation : KDOQI clinical practice guideline for diabetes and CKD: 2012 update. Am J Kidney Dis 60: 850–886, 2012 - PubMed
-
- American Diabetes Association : Cardiovascular disease and risk management, section 8. In standards of medical care in diabetes-2016. Diabetes Care 39(Suppl 1): S60–S71, 2016 - PubMed
-
- United Stated Renal Data System (USRDS). 2016 Annual Data Report, Vol 1, CKD, Chapter 1. Available at: http://www.usrds.org. Accessed January 3, 2017
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

