A direct method for the N-tetraalkylation of azamacrocycles
- PMID: 28144313
- PMCID: PMC5238601
- DOI: 10.3762/bjoc.12.239
A direct method for the N-tetraalkylation of azamacrocycles
Abstract
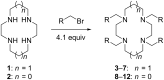
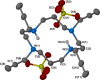
An efficient protocol for the direct synthesis of N-tetraalkylated derivatives of the azamacrocycles cyclam and cyclen has been developed, using a partially miscible aqueous-organic solvent system with propargyl bromide, benzyl bromide, and related halides. The method works most effectively when the reaction mixture is shaken, not stirred. A crystal structure of the N-tetrapropargyl cyclam derivative 1,4,8,11-tetra(prop-2-yn-1-yl)-1,4,8,11-tetraazacyclotetradecane diperchlorate is reported.
Keywords: N-alkylation; azamacrocycles; biphasic system; cyclam; cyclen.
Figures
Similar articles
-
Mono- and Bis-Alkylation of Glyoxal-Bridged Tetraazamacrocycles Using Mechanochemistry.J Org Chem. 2016 Feb 5;81(3):890-8. doi: 10.1021/acs.joc.5b02464. Epub 2016 Jan 21. J Org Chem. 2016. PMID: 26670783
-
Monopicolinate cyclen and cyclam derivatives for stable copper(II) complexation.Inorg Chem. 2012 Jun 18;51(12):6916-27. doi: 10.1021/ic300784v. Epub 2012 May 31. Inorg Chem. 2012. PMID: 22651379
-
Mononuclear (nitrido)iron(V) and (oxo)iron(IV) complexes via photolysis of [(cyclam-acetato)FeIII(N3)]+ and ozonolysis of [(cyclam-acetato)FeIII(O3SCF3)]+ in water/acetone mixtures.Inorg Chem. 2000 Nov 13;39(23):5306-17. doi: 10.1021/ic0005238. Inorg Chem. 2000. PMID: 11187471
-
Syntheses of 8-quinolinolatocobalt(III) complexes containing cyclen based auxiliary ligands as models for hypoxia-activated prodrugs.Dalton Trans. 2010 Dec 28;39(48):11535-50. doi: 10.1039/c0dt01142h. Epub 2010 Nov 22. Dalton Trans. 2010. PMID: 21103540
-
A mixed-valence manganese(III)-manganese(IV) di-mu-oxo complex, [(cyclam)MnO]2(ClO4)2(NO3).Acta Crystallogr C. 2001 Aug;57(Pt 8):911-3. doi: 10.1107/s0108270101008617. Epub 2001 Aug 9. Acta Crystallogr C. 2001. PMID: 11498610
Cited by
-
Arabinosylation of cell wall extensin is required for the directional response to salinity in roots.Plant Cell. 2024 Sep 3;36(9):3328-3343. doi: 10.1093/plcell/koae135. Plant Cell. 2024. PMID: 38691576 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
