Measuring Deformation in the Mouse Optic Nerve Head and Peripapillary Sclera
- PMID: 28146237
- PMCID: PMC5295769
- DOI: 10.1167/iovs.16-20620
Measuring Deformation in the Mouse Optic Nerve Head and Peripapillary Sclera
Abstract
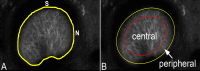
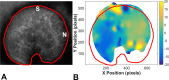
Purpose: To develop an ex vivo explant system using multiphoton microscopy and digital volume correlation to measure the full-field deformation response to intraocular pressure (IOP) change in the peripapillary sclera (PPS) and in the optic nerve head (ONH) astrocytic structure.
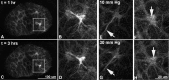
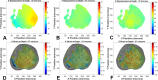
Methods: Green fluorescent protein (GFP)-glutamate transporter-GLT1 (GLT1/GFP) mouse eyes were explanted and imaged with a laser-scanning microscope under controlled inflation. Images were analyzed for regional strains and changes in astrocytic lamina and PPS shape. Astrocyte volume fraction in seven control GLT1/GFP mice was measured. The level of fluorescence of GFP fluorescent astrocytes was compared with glial fibrillary acidic protein (GFAP) labeled astrocytes using immunohistochemistry.
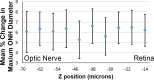
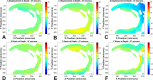
Results: The ONH astrocytic structure remained stable during 3 hours in explants. Control strain-globally, in the central one-half or two-thirds of the astrocytic lamina-was significantly greater in the nasal-temporal direction than in the inferior-superior or anterior-posterior directions (each P≤ 0.03, mixed models). The PPS opening (perimeter) in normal eye explants also became wider nasal-temporally than superior-inferiorly during inflation from 10 to 30 mm Hg (P = 0.0005). After 1 to 3 days of chronic IOP elevation, PPS area was larger than in control eyes (P = 0.035), perimeter elongation was 37% less than controls, and global nasal-temporal strain was significantly less than controls (P = 0.007). Astrocyte orientation was altered by chronic IOP elevation, with processes redirected toward the longitudinal axis of the optic nerve.
Conclusions: The explant inflation test measures the strain response of the mouse ONH to applied IOP. Initial studies indicate regional differences in response to both acute and chronic IOP elevation within the ONH region.
Figures


Comment in
-
Ex Vivo Imaging of the Murine Optic Nerve Head.Invest Ophthalmol Vis Sci. 2017 Feb 1;58(2):734. doi: 10.1167/iovs.17-21422. Invest Ophthalmol Vis Sci. 2017. PMID: 28152143 No abstract available.
Similar articles
-
Age-Related Changes in Quantitative Strain of Mouse Astrocytic Lamina Cribrosa and Peripapillary Sclera Using Confocal Microscopy in an Explant Model.Invest Ophthalmol Vis Sci. 2018 Oct 1;59(12):5157-5166. doi: 10.1167/iovs.18-25111. Invest Ophthalmol Vis Sci. 2018. PMID: 30372742 Free PMC article.
-
Regional Deformation of the Optic Nerve Head and Peripapillary Sclera During IOP Elevation.Invest Ophthalmol Vis Sci. 2018 Jul 2;59(8):3779-3788. doi: 10.1167/iovs.18-24462. Invest Ophthalmol Vis Sci. 2018. PMID: 30046819 Free PMC article.
-
A mouse ocular explant model that enables the study of living optic nerve head events after acute and chronic intraocular pressure elevation: Focusing on retinal ganglion cell axons and mitochondria.Exp Eye Res. 2017 Jul;160:106-115. doi: 10.1016/j.exer.2017.04.003. Epub 2017 Apr 14. Exp Eye Res. 2017. PMID: 28414059 Free PMC article.
-
Biological aspects of axonal damage in glaucoma: A brief review.Exp Eye Res. 2017 Apr;157:5-12. doi: 10.1016/j.exer.2017.02.006. Epub 2017 Feb 20. Exp Eye Res. 2017. PMID: 28223179 Free PMC article. Review.
-
The optic nerve head as a biomechanical structure: a new paradigm for understanding the role of IOP-related stress and strain in the pathophysiology of glaucomatous optic nerve head damage.Prog Retin Eye Res. 2005 Jan;24(1):39-73. doi: 10.1016/j.preteyeres.2004.06.001. Prog Retin Eye Res. 2005. PMID: 15555526 Review.
Cited by
-
Pressure-Induced Changes in Astrocyte GFAP, Actin, and Nuclear Morphology in Mouse Optic Nerve.Invest Ophthalmol Vis Sci. 2020 Sep 1;61(11):14. doi: 10.1167/iovs.61.11.14. Invest Ophthalmol Vis Sci. 2020. PMID: 32910133 Free PMC article.
-
The Mechanisms of Neuroprotection by Topical Rho Kinase Inhibition in Experimental Mouse Glaucoma and Optic Neuropathy.Invest Ophthalmol Vis Sci. 2024 Nov 4;65(13):43. doi: 10.1167/iovs.65.13.43. Invest Ophthalmol Vis Sci. 2024. PMID: 39565302 Free PMC article.
-
Analysis of 2-dimensional regional differences in the peripapillary scleral fibroblast cytoskeleton of normotensive and hypertensive mouse eyes.Sci Rep. 2025 Jul 1;15(1):21207. doi: 10.1038/s41598-025-08251-4. Sci Rep. 2025. PMID: 40595239 Free PMC article.
-
Simultaneous Assessment of the Whole Eye Biomechanics Using Ultrasonic Elastography.IEEE Trans Biomed Eng. 2023 Apr;70(4):1310-1317. doi: 10.1109/TBME.2022.3215498. Epub 2023 Mar 22. IEEE Trans Biomed Eng. 2023. PMID: 36260593 Free PMC article.
-
Morphometric and Microstructural Changes During Murine Retinal Development Characterized Using In Vivo Optical Coherence Tomography.Invest Ophthalmol Vis Sci. 2021 Oct 4;62(13):20. doi: 10.1167/iovs.62.13.20. Invest Ophthalmol Vis Sci. 2021. PMID: 34698774 Free PMC article.
References
-
- Bengtsson B,, Heijl A. Diurnal IOP fluctuation: not an independent risk factor for glaucomatous visual field loss in high-risk ocular hypertension. Graefe's Arch Clin Exper Ophthalmol. 2005; 243: 513–518. - PubMed
-
- Nouri-Mahdavi K,, Hoffman D,, Coleman A. Predictive factors for glaucomatous visual field progression in the Advanced Glaucoma Intervention Study. Ophthalmology. 2004; 111: 1627–1635. - PubMed
-
- De Moraes CG,, Juthani VJ,, Liebmann JM. Risk factors for visual field progression in treated glaucoma. Arch Ophthalmol. 2011; 129: 562–568. - PubMed
-
- Burgoyne CF,, Downs JC,, Bellezza AJ,, Suh JK,, Hart RT. The optic nerve head as a biomechanical structure: a new paradigm for understanding the role of IOP-related stress and strain in the pathophysiology of glaucomatous optic nerve head damage. Prog Retin Eye Res. 2005; 24: 39–73. - PubMed
-
- Ethier CR. Scleral biomechanics and glaucoma–a connection? Can J Ophthalmol. 2006; 41: 9–11. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

