Stability of Circulating Blood-Based MicroRNAs - Pre-Analytic Methodological Considerations
- PMID: 28151938
- PMCID: PMC5289450
- DOI: 10.1371/journal.pone.0167969
Stability of Circulating Blood-Based MicroRNAs - Pre-Analytic Methodological Considerations
Abstract
Background and aim: The potential of microRNAs (miRNA) as non-invasive diagnostic, prognostic, and predictive biomarkers, as well as therapeutic targets, has recently been recognized. Previous studies have highlighted the importance of consistency in the methodology used, but to our knowledge, no study has described the methodology of sample preparation and storage systematically with respect to miRNAs as blood biomarkers. The aim of this study was to investigate the stability of miRNAs in blood under various relevant clinical and research conditions: different collection tubes, storage at different temperatures, physical disturbance, as well as serial freeze-thaw cycles.
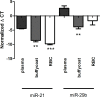
Methods: Blood samples were collected from 12 healthy donors into different collection tubes containing anticoagulants, including EDTA, citrate and lithium-heparin, as well as into serum collection tubes. MiRNA stability was evaluated by measuring expression changes of miR-1, miR-21 and miR-29b at different conditions: varying processing time of whole blood (up to 72 hours (h)), long-term storage (9 months at -80°C), physical disturbance (1 and 8 h), as well as in a series of freeze/thaw cycles (1 and 4 times).
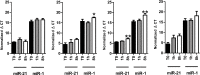
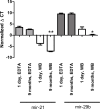
Results: Different collection tubes revealed comparable concentrations of miR-1, miR-21 and miR-29b. Tubes with lithium-heparin were found unsuitable for miRNA quantification. MiRNA levels were stable for at least 24 h at room temperature in whole blood, while separated fractions did show alterations within 24 h. There were significant changes in the miR-21 and miR-29b levels after 72 h incubation of whole blood at room temperature (p<0.01 for both). Both miR-1 and miR-21 showed decreased levels after physical disturbance for 8 h in separated plasma and miR-1 in serum whole blood, while after 1 h of disturbance no changes were observed. Storage of samples at -80°C extended the miRNA stability remarkably, however, miRNA levels in long-term stored (9 months) whole blood samples were significantly changed, which is in contrast to the plasma samples, where miR-21 or miR-29b levels were found to be stable. Repetitive (n = 4) freeze-thaw cycles resulted in a significant reduction of miRNA concentration both in plasma and serum samples.
Conclusion: This study highlights the importance of proper and systematic sample collection and preparation when measuring circulating miRNAs, e.g., in context of clinical trials. We demonstrated that the type of collection tubes, preparation, handling and storage of samples should be standardized to avoid confounding variables influencing the results.
Conflict of interest statement
This study received funding from the Novo Nordic Foundation. There are no patents, products in development or marketed products to declare. This does not alter our adherence to all the PLOS ONE policies on sharing data and materials.
Figures
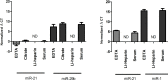


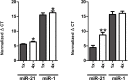
Similar articles
-
Impact of long-term storage and freeze-thawing on eight circulating microRNAs in plasma samples.PLoS One. 2020 Jan 14;15(1):e0227648. doi: 10.1371/journal.pone.0227648. eCollection 2020. PLoS One. 2020. PMID: 31935258 Free PMC article.
-
Red blood cells as potential materials for microRNA biomarker study: overcoming heparin-related challenges.Am J Physiol Heart Circ Physiol. 2024 Nov 1;327(5):H1296-H1302. doi: 10.1152/ajpheart.00609.2024. Epub 2024 Oct 18. Am J Physiol Heart Circ Physiol. 2024. PMID: 39422364
-
Towards Clinical Applications of Blood-Borne miRNA Signatures: The Influence of the Anticoagulant EDTA on miRNA Abundance.PLoS One. 2015 Nov 23;10(11):e0143321. doi: 10.1371/journal.pone.0143321. eCollection 2015. PLoS One. 2015. PMID: 26599228 Free PMC article.
-
Variability in, variability out: best practice recommendations to standardize pre-analytical variables in the detection of circulating and tissue microRNAs.Clin Chem Lab Med. 2017 May 1;55(5):608-621. doi: 10.1515/cclm-2016-0471. Clin Chem Lab Med. 2017. PMID: 28306519 Review.
-
Methodological challenges in utilizing miRNAs as circulating biomarkers.J Cell Mol Med. 2014 Mar;18(3):371-90. doi: 10.1111/jcmm.12236. Epub 2014 Feb 18. J Cell Mol Med. 2014. PMID: 24533657 Free PMC article. Review.
Cited by
-
Epigenetic analyses in forensic medicine: future and challenges.Int J Legal Med. 2024 May;138(3):701-719. doi: 10.1007/s00414-024-03165-8. Epub 2024 Jan 20. Int J Legal Med. 2024. PMID: 38242965 Free PMC article. Review.
-
Reproducibility of the Infinium methylationEPIC BeadChip assay using low DNA amounts.Epigenetics. 2022 Dec;17(12):1636-1645. doi: 10.1080/15592294.2022.2051861. Epub 2022 Mar 31. Epigenetics. 2022. PMID: 35356867 Free PMC article.
-
Microarray analysis identifies coding and non-coding RNA markers of liver injury in whole body irradiated mice.Sci Rep. 2023 Jan 5;13(1):200. doi: 10.1038/s41598-022-26784-w. Sci Rep. 2023. PMID: 36604457 Free PMC article.
-
Differential Circulating MicroRNA Expression in Age-Related Macular Degeneration.Int J Mol Sci. 2021 Nov 15;22(22):12321. doi: 10.3390/ijms222212321. Int J Mol Sci. 2021. PMID: 34830203 Free PMC article.
-
Comparison of miRNA quantitation by Nanostring in serum and plasma samples.PLoS One. 2017 Dec 6;12(12):e0189165. doi: 10.1371/journal.pone.0189165. eCollection 2017. PLoS One. 2017. PMID: 29211799 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

