Choline and methionine differentially alter methyl carbon metabolism in bovine neonatal hepatocytes
- PMID: 28152052
- PMCID: PMC5289486
- DOI: 10.1371/journal.pone.0171080
Choline and methionine differentially alter methyl carbon metabolism in bovine neonatal hepatocytes
Abstract
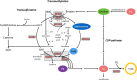
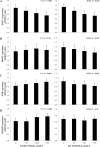
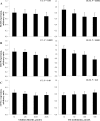
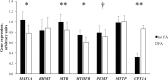
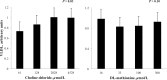
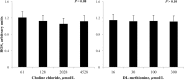
Intersections in hepatic methyl group metabolism pathways highlights potential competition or compensation of methyl donors. The objective of this experiment was to examine the expression of genes related to methyl group transfer and lipid metabolism in response to increasing concentrations of choline chloride (CC) and DL-methionine (DLM) in primary neonatal hepatocytes that were or were not exposed to fatty acids (FA). Primary hepatocytes isolated from 4 neonatal Holstein calves were maintained as monolayer cultures for 24 h before treatment with CC (61, 128, 2028, and 4528 μmol/L) and DLM (16, 30, 100, 300 μmol/L), with or without a 1 mmol/L FA cocktail in a factorial arrangement. After 24 h of treatment, media was collected for quantification of reactive oxygen species (ROS) and very low-density lipoprotein (VLDL), and cell lysates were collected for quantification of gene expression. No interactions were detected between CC, DLM, or FA. Both CC and DLM decreased the expression of methionine adenosyltransferase 1A (MAT1A). Increasing CC did not alter betaine-homocysteine S-methyltranferase (BHMT) but did increase 5-methyltetrahydrofolate-homocysteine methyltransferase (MTR) and methylenetetrahydrofolate reductase (MTHFR) expression. Increasing DLM decreased expression of BHMT and MTR, but did not affect MTHFR. Expression of both phosphatidylethanolamine N-methyltransferase (PEMT) and microsomal triglyceride transfer protein (MTTP) were decreased by increasing CC and DLM, while carnitine palmitoyltransferase 1A (CPT1A) was unaffected by either. Treatment with FA decreased the expression of MAT1A, MTR, MTHFR and tended to decrease PEMT but did not affect BHMT and MTTP. Treatment with FA increased CPT1A expression. Increasing CC increased secretion of VLDL and decreased the accumulation of ROS in media. Within neonatal bovine hepatocytes, choline and methionine differentially regulate methyl carbon pathways and suggest that choline may play a critical role in donating methyl groups to support methionine regeneration. Stimulating VLDL export and decreasing ROS accumulation suggests that increasing CC is hepato-protective.
Conflict of interest statement
HMW has received honorarium for presenting results of this research at conferences or technical meetings organized or sponsored by Balchem Corporation. TLC has declared no competing interests exist. This does not alter adherence to PLOS ONE policies on sharing data and materials.
Figures
Similar articles
-
Palmitate and pyruvate carbon flux in response to choline and methionine in bovine neonatal hepatocytes.Sci Rep. 2020 Nov 5;10(1):19078. doi: 10.1038/s41598-020-75956-z. Sci Rep. 2020. PMID: 33154483 Free PMC article.
-
Glucose metabolism is differentially altered by choline and methionine in bovine neonatal hepatocytes.PLoS One. 2019 May 29;14(5):e0217160. doi: 10.1371/journal.pone.0217160. eCollection 2019. PLoS One. 2019. PMID: 31141525 Free PMC article.
-
The effect of increasing concentrations of dl-methionine and 2-hydroxy-4-(methylthio) butanoic acid on hepatic genes controlling methionine regeneration and gluconeogenesis.J Dairy Sci. 2016 Oct;99(10):8451-8460. doi: 10.3168/jds.2016-11312. Epub 2016 Jul 27. J Dairy Sci. 2016. PMID: 27474977
-
Pleiotropic effects of methionine adenosyltransferases deregulation as determinants of liver cancer progression and prognosis.J Hepatol. 2013 Oct;59(4):830-41. doi: 10.1016/j.jhep.2013.04.031. Epub 2013 May 7. J Hepatol. 2013. PMID: 23665184 Review.
-
High Folate, Perturbed One-Carbon Metabolism and Gestational Diabetes Mellitus.Nutrients. 2022 Sep 22;14(19):3930. doi: 10.3390/nu14193930. Nutrients. 2022. PMID: 36235580 Free PMC article. Review.
Cited by
-
Palmitate and pyruvate carbon flux in response to choline and methionine in bovine neonatal hepatocytes.Sci Rep. 2020 Nov 5;10(1):19078. doi: 10.1038/s41598-020-75956-z. Sci Rep. 2020. PMID: 33154483 Free PMC article.
-
Deconvolution of the epigenetic age discloses distinct inter-personal variability in epigenetic aging patterns.Epigenetics Chromatin. 2022 Mar 7;15(1):9. doi: 10.1186/s13072-022-00441-y. Epigenetics Chromatin. 2022. PMID: 35255955 Free PMC article.
-
Liver and Muscle Transcriptomes Differ in Mid-Lactation Cows Divergent in Feed Efficiency in the Presence or Absence of Supplemental Rumen-Protected Choline.Metabolites. 2023 Sep 19;13(9):1023. doi: 10.3390/metabo13091023. Metabolites. 2023. PMID: 37755303 Free PMC article.
-
Multifaceted role of one-carbon metabolism on immunometabolic control and growth during pregnancy, lactation and the neonatal period in dairy cattle.J Anim Sci Biotechnol. 2021 Feb 4;12(1):27. doi: 10.1186/s40104-021-00547-5. J Anim Sci Biotechnol. 2021. PMID: 33536062 Free PMC article. Review.
-
Glucose metabolism is differentially altered by choline and methionine in bovine neonatal hepatocytes.PLoS One. 2019 May 29;14(5):e0217160. doi: 10.1371/journal.pone.0217160. eCollection 2019. PLoS One. 2019. PMID: 31141525 Free PMC article.
References
-
- Xue GP, Snoswell AM. Comparative studies on the methionine synthesis in sheep and rat tissues. Comp. Biochem. Phys. 1985; 80: 489–94. - PubMed
-
- Sharma BK, Erdman RA. In Vitro Degradation of Choline from Selected Foodstuffs and Choline Supplements. J. Dairy Sci. 1989; 72: 2772–6. - PubMed
-
- de Veth MJ, Artegoitia VM, Campagna SR, Lapierre H, Harte F, Girard CL. Choline absorption and evaluation of bioavailability markers when supplementing choline to lactating dairy cows. J. Dairy Sci. 2016; 12: 1–13. - PubMed
-
- Herrick KJ, Davidson JA, Valdez FR, Christofferson MJ, Schuling SE. Bioavailability of rumen protected choline sources when supplemented at different concentrations. J. Dairy Sci. 2014; 97(Suppl. 1): 1896(Abstrt.).
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

