β1 integrin signaling promotes neuronal migration along vascular scaffolds in the post-stroke brain
- PMID: 28153772
- PMCID: PMC5474439
- DOI: 10.1016/j.ebiom.2017.01.005
β1 integrin signaling promotes neuronal migration along vascular scaffolds in the post-stroke brain
Abstract
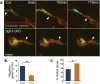
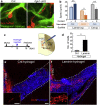
Cerebral ischemic stroke is a main cause of chronic disability. However, there is currently no effective treatment to promote recovery from stroke-induced neurological symptoms. Recent studies suggest that after stroke, immature neurons, referred to as neuroblasts, generated in a neurogenic niche, the ventricular-subventricular zone, migrate toward the injured area, where they differentiate into mature neurons. Interventions that increase the number of neuroblasts distributed at and around the lesion facilitate neuronal repair in rodent models for ischemic stroke, suggesting that promoting neuroblast migration in the post-stroke brain could improve efficient neuronal regeneration. To move toward the lesion, neuroblasts form chain-like aggregates and migrate along blood vessels, which are thought to increase their migration efficiency. However, the molecular mechanisms regulating these migration processes are largely unknown. Here we studied the role of β1-class integrins, transmembrane receptors for extracellular matrix proteins, in these migrating neuroblasts. We found that the neuroblast chain formation and blood vessel-guided migration critically depend on β1 integrin signaling. β1 integrin facilitated the adhesion of neuroblasts to laminin and the efficient translocation of their soma during migration. Moreover, artificial laminin-containing scaffolds promoted neuroblast chain formation and migration toward the injured area. These data suggest that laminin signaling via β1 integrin supports vasculature-guided neuronal migration to efficiently supply neuroblasts to injured areas. This study also highlights the importance of vascular scaffolds for cell migration in development and regeneration.
Keywords: Blood vessel; Chain migration; Laminin; Stroke; Vasculature-guided migration; β1 integrin.
Copyright © 2017 3-V Biosciences. Published by Elsevier B.V. All rights reserved.
Figures



References
-
- Ajioka I., Jinnou H., Okada K., Sawada M., Saitoh S., Sawamoto K. Enhancement of neuroblast migration into the injured cerebral cortex using laminin-containing porous sponge. Tissue Eng. A. 2015;21:193–201. - PubMed
-
- Bussmann J., Bos F.L., Urasaki A., Kawakami K., Duckers H.J., Schulte-Merker S. Arteries provide essential guidance cues for lymphatic endothelial cells in the zebrafish trunk. Development. 2010;137:2653–2657. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

