Cdon deficiency causes cardiac remodeling through hyperactivation of WNT/β-catenin signaling
- PMID: 28154134
- PMCID: PMC5338397
- DOI: 10.1073/pnas.1615105114
Cdon deficiency causes cardiac remodeling through hyperactivation of WNT/β-catenin signaling
Abstract
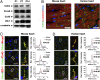
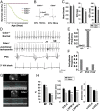
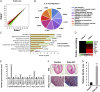
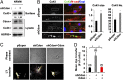
On pathological stress, Wnt signaling is reactivated and induces genes associated with cardiac remodeling and fibrosis. We have previously shown that a cell surface receptor Cdon (cell-adhesion associated, oncogene regulated) suppresses Wnt signaling to promote neuronal differentiation however its role in heart is unknown. Here, we demonstrate a critical role of Cdon in cardiac function and remodeling. Cdon is expressed and predominantly localized at intercalated disk in both mouse and human hearts. Cdon-deficient mice develop cardiac dysfunction including reduced ejection fraction and ECG abnormalities. Cdon-/- hearts exhibit increased fibrosis and up-regulation of genes associated with cardiac remodeling and fibrosis. Electrical remodeling was demonstrated by up-regulation and mislocalization of the gap junction protein, Connexin 43 (Cx43) in Cdon-/- hearts. In agreement with altered Cx43 expression, functional analysis both using Cdon-/- cardiomyocytes and shRNA-mediated knockdown in rat cardiomyocytes shows aberrant gap junction activities. Analysis of the underlying mechanism reveals that Cdon-/- hearts exhibit hyperactive Wnt signaling as evident by β-catenin accumulation and Axin2 up-regulation. On the other hand, the treatment of rat cardiomyocytes with a Wnt activator TWS119 reduces Cdon levels and aberrant Cx43 activities, similarly to Cdon-deficient cardiomyocytes, suggesting a negative feedback between Cdon and Wnt signaling. Finally, inhibition of Wnt/β-catenin signaling by XAV939, IWP2 or dickkopf (DKK)1 prevented Cdon depletion-induced up-regulation of collagen 1a and Cx43. Taken together, these results demonstrate that Cdon deficiency causes hyperactive Wnt signaling leading to aberrant intercellular coupling and cardiac fibrosis. Cdon exhibits great potential as a target for the treatment of cardiac fibrosis and cardiomyopathy.
Keywords: Cdon; Wnt; cardiac fibrosis; connexin 43; gap junction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Wnt-1 regulation of connexin43 in cardiac myocytes.J Clin Invest. 2000 Jan;105(2):161-71. doi: 10.1172/JCI7798. J Clin Invest. 2000. PMID: 10642594 Free PMC article.
-
LRP6 acts as a scaffold protein in cardiac gap junction assembly.Nat Commun. 2016 Jun 2;7:11775. doi: 10.1038/ncomms11775. Nat Commun. 2016. PMID: 27250245 Free PMC article.
-
Rapid electrical stimulation causes alterations in cardiac intercellular junction proteins of cardiomyocytes.Am J Physiol Heart Circ Physiol. 2014 May;306(9):H1324-33. doi: 10.1152/ajpheart.00653.2013. Epub 2014 Mar 7. Am J Physiol Heart Circ Physiol. 2014. PMID: 24610920
-
[Remodeling of cardiac gap junctions and arrhythmias].Sheng Li Xue Bao. 2011 Dec 25;63(6):586-92. Sheng Li Xue Bao. 2011. PMID: 22193455 Review. Chinese.
-
Alterations in cardiac connexin expression in cardiomyopathies.Adv Cardiol. 2006;42:228-242. doi: 10.1159/000092572. Adv Cardiol. 2006. PMID: 16646594 Review.
Cited by
-
Integrated multiomics of pressure overload in the human heart prioritizes targets relevant to heart failure.Nat Commun. 2025 Jul 26;16(1):6889. doi: 10.1038/s41467-025-62201-2. Nat Commun. 2025. PMID: 40715126 Free PMC article.
-
Increased GIRK channel activity prevents arrhythmia in mice with heart failure by enhancing ventricular repolarization.Sci Rep. 2023 Dec 18;13(1):22479. doi: 10.1038/s41598-023-50088-2. Sci Rep. 2023. PMID: 38110503 Free PMC article.
-
Inducible Prmt1 ablation in adult vascular smooth muscle leads to contractile dysfunction and aortic dissection.Exp Mol Med. 2021 Oct;53(10):1569-1579. doi: 10.1038/s12276-021-00684-x. Epub 2021 Oct 11. Exp Mol Med. 2021. PMID: 34635781 Free PMC article.
-
Activation of hedgehog signaling in mesenchymal stem cells induces cartilage and bone tumor formation via Wnt/β-Catenin.Elife. 2019 Sep 4;8:e50208. doi: 10.7554/eLife.50208. Elife. 2019. PMID: 31482846 Free PMC article.
-
Inhibition of PRMT6 reduces neomycin-induced inner ear hair cell injury through the restraint of FoxG1 arginine methylation.Inflamm Res. 2022 Mar;71(3):309-320. doi: 10.1007/s00011-022-01541-x. Epub 2022 Feb 21. Inflamm Res. 2022. PMID: 35190853
References
-
- Katz AM. Cardiomyopathy of overload. A major determinant of prognosis in congestive heart failure. N Engl J Med. 1990;322(2):100–110. - PubMed
-
- Mozaffarian D, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee Heart disease and stroke statistics--2015 update: A report from the American Heart Association. Circulation. 2015;131(4):e29–e322. - PubMed
-
- Creemers EE, Pinto YM. Molecular mechanisms that control interstitial fibrosis in the pressure-overloaded heart. Cardiovasc Res. 2011;89(2):265–272. - PubMed
-
- Flesch M, et al. Sarcoplasmic reticulum Ca2+ATPase and phospholamban mRNA and protein levels in end-stage heart failure due to ischemic or dilated cardiomyopathy. J Mol Med (Berl) 1996;74(6):321–332. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

