WRN is recruited to damaged telomeres via its RQC domain and tankyrase1-mediated poly-ADP-ribosylation of TRF1
- PMID: 28158503
- PMCID: PMC5397154
- DOI: 10.1093/nar/gkx065
WRN is recruited to damaged telomeres via its RQC domain and tankyrase1-mediated poly-ADP-ribosylation of TRF1
Abstract
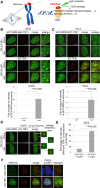
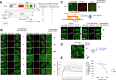
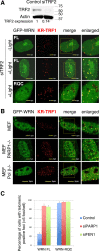
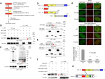
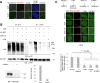
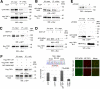
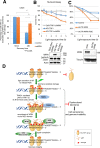
Werner syndrome (WS) is a progeroid-like syndrome caused by WRN gene mutations. WS cells exhibit shorter telomere length compared to normal cells, but it is not fully understood how WRN deficiency leads directly to telomere dysfunction. By generating localized telomere-specific DNA damage in a real-time fashion and a dose-dependent manner, we found that the damage response of WRN at telomeres relies on its RQC domain, which is different from the canonical damage response at genomic sites via its HRDC domain. We showed that in addition to steady state telomere erosion, WRN depleted cells are also sensitive to telomeric damage. WRN responds to site-specific telomeric damage via its RQC domain, interacting at Lysine 1016 and Phenylalanine1037 with the N-terminal acidic domain of the telomere shelterin protein TRF1 and demonstrating a novel mechanism for WRN's role in telomere protection. We also found that tankyrase1-mediated poly-ADP-ribosylation of TRF1 is important for both the interaction between WRN and TRF1 and the damage recruitment of WRN to telomeres. Mutations of potential tankyrase1 ADP-ribosylation sites within the RGCADG motif of TRF1 strongly diminish the interaction with WRN and the damage response of WRN only at telomeres. Taken together, our results reveal a novel mechanism as to how WRN protects telomere integrity from damage and telomere erosion.
© The Author(s) 2017. Published by Oxford University Press on behalf of Nucleic Acids Research.
Figures
Similar articles
-
Tankyrase1-mediated poly(ADP-ribosyl)ation of TRF1 maintains cell survival after telomeric DNA damage.Nucleic Acids Res. 2017 Apr 20;45(7):3906-3921. doi: 10.1093/nar/gkx083. Nucleic Acids Res. 2017. PMID: 28160604 Free PMC article.
-
The role of the poly(ADP-ribose) polymerase tankyrase1 in telomere length control by the TRF1 component of the shelterin complex.J Biol Chem. 2007 Aug 3;282(31):22662-7. doi: 10.1074/jbc.M702620200. Epub 2007 Jun 8. J Biol Chem. 2007. PMID: 17561506
-
The Werner syndrome helicase and exonuclease cooperate to resolve telomeric D loops in a manner regulated by TRF1 and TRF2.Mol Cell. 2004 Jun 18;14(6):763-74. doi: 10.1016/j.molcel.2004.05.023. Mol Cell. 2004. PMID: 15200954
-
Pathways and functions of the Werner syndrome protein.Mech Ageing Dev. 2005 Jan;126(1):79-86. doi: 10.1016/j.mad.2004.09.011. Mech Ageing Dev. 2005. PMID: 15610765 Review.
-
Werner Syndrome Protein and DNA Replication.Int J Mol Sci. 2018 Nov 2;19(11):3442. doi: 10.3390/ijms19113442. Int J Mol Sci. 2018. PMID: 30400178 Free PMC article. Review.
Cited by
-
Research on Werner Syndrome: Trends from Past to Present and Future Prospects.Genes (Basel). 2022 Oct 6;13(10):1802. doi: 10.3390/genes13101802. Genes (Basel). 2022. PMID: 36292687 Free PMC article. Review.
-
Dynamics of TRF1 organizing a single human telomere.Nucleic Acids Res. 2021 Jan 25;49(2):760-775. doi: 10.1093/nar/gkaa1222. Nucleic Acids Res. 2021. PMID: 33347580 Free PMC article.
-
Family-based whole-exome sequencing identifies rare variants potentially related to cutaneous melanoma predisposition in Brazilian melanoma-prone families.PLoS One. 2022 Jan 27;17(1):e0262419. doi: 10.1371/journal.pone.0262419. eCollection 2022. PLoS One. 2022. PMID: 35085295 Free PMC article.
-
DNA damage and mitochondria in cancer and aging.Carcinogenesis. 2020 Dec 31;41(12):1625-1634. doi: 10.1093/carcin/bgaa114. Carcinogenesis. 2020. PMID: 33146705 Free PMC article. Review.
-
Cerebral Haemorrhage in a Young Patient With Atypical Werner Syndrome Due to Mutations in LMNA.Front Endocrinol (Lausanne). 2018 Aug 3;9:433. doi: 10.3389/fendo.2018.00433. eCollection 2018. Front Endocrinol (Lausanne). 2018. PMID: 30123186 Free PMC article.
References
-
- Cooke M.S., Evans M.D., Dizdaroglu M., Lunec J.. Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J. 2003; 17:1195–1214. - PubMed
-
- Richter T., von Zglinicki T.. A continuous correlation between oxidative stress and telomere shortening in fibroblasts. Exp. Gerontol. 2007; 42:1039–1042. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

