NLRX1 Regulates Effector and Metabolic Functions of CD4+ T Cells
- PMID: 28159898
- PMCID: PMC5340590
- DOI: 10.4049/jimmunol.1601547
NLRX1 Regulates Effector and Metabolic Functions of CD4+ T Cells
Abstract
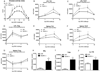
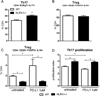
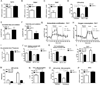
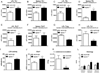
Nucleotide oligomerization domain-like receptor X1 (NLRX1) has been implicated in viral response, cancer progression, and inflammatory disorders; however, its role as a dual modulator of CD4+ T cell function and metabolism has not been defined. The loss of NLRX1 results in increased disease severity, populations of Th1 and Th17 cells, and inflammatory markers (IFN-γ, TNF-α, and IL-17) in mice with dextran sodium sulfate-induced colitis. To further characterize this phenotype, we used in vitro CD4+ T cell-differentiation assays and show that NLRX1-deficient T cells have a greater ability to differentiate into an inflammatory phenotype and possess greater proliferation rates. Further, NLRX1-/- cells have a decreased responsiveness to immune checkpoint pathways and greater rates of lactate dehydrogenase activity. When metabolic effects of the knockout are impaired, NLRX1-deficient cells do not display significant differences in differentiation or proliferation. To confirm the role of NLRX1 specifically in T cells, we used an adoptive-transfer model of colitis. Rag2-/- mice receiving NLRX1-/- naive or effector T cells experienced increased disease activity and effector T cell populations, whereas no differences were observed between groups receiving wild-type or NLRX1-/- regulatory T cells. Metabolic effects of NLRX1 deficiency are observed in a CD4-specific knockout of NLRX1 within a Citrobacter rodentium model of colitis. The aerobic glycolytic preference in NLRX1-/- effector T cells is combined with a decreased sensitivity to immunosuppressive checkpoint pathways to provide greater proliferative capabilities and an inflammatory phenotype bias leading to increased disease severity.
Copyright © 2017 by The American Association of Immunologists, Inc.
Conflict of interest statement
Figures



Similar articles
-
NLRX1 Modulates Immunometabolic Mechanisms Controlling the Host-Gut Microbiota Interactions during Inflammatory Bowel Disease.Front Immunol. 2018 Feb 26;9:363. doi: 10.3389/fimmu.2018.00363. eCollection 2018. Front Immunol. 2018. PMID: 29535731 Free PMC article.
-
The IL-21/IL-21R signaling axis regulates CD4+ T-cell responsiveness to IL-12 to promote bacterial-induced colitis.J Leukoc Biol. 2024 Oct 1;116(4):726-737. doi: 10.1093/jleuko/qiae069. J Leukoc Biol. 2024. PMID: 38498592 Free PMC article.
-
IL-33 induces both regulatory B cells and regulatory T cells in dextran sulfate sodium-induced colitis.Int Immunopharmacol. 2017 May;46:38-47. doi: 10.1016/j.intimp.2017.02.006. Epub 2017 Feb 28. Int Immunopharmacol. 2017. PMID: 28258042
-
Cytokines in experimental colitis.Clin Exp Immunol. 1999 Dec;118(3):337-9. doi: 10.1046/j.1365-2249.1999.01088.x. Clin Exp Immunol. 1999. PMID: 10594548 Free PMC article. Review. No abstract available.
-
Effector and regulatory CD4+ T cell function in a murine model of Helicobacter hepaticus-induced colitis.J Pediatr Gastroenterol Nutr. 2005 Apr;40 Suppl 1:S35-6. doi: 10.1097/00005176-200504001-00021. J Pediatr Gastroenterol Nutr. 2005. PMID: 15805844 Review. No abstract available.
Cited by
-
Identification of new regulatory genes through expression pattern analysis of a global RNA-seq dataset from a Helicobacter pylori co-culture system.Sci Rep. 2020 Jul 13;10(1):11506. doi: 10.1038/s41598-020-68439-8. Sci Rep. 2020. PMID: 32661418 Free PMC article.
-
LRR domain of NLRX1 protein delivery by dNP2 inhibits T cell functions and alleviates autoimmune encephalomyelitis.Theranostics. 2020 Feb 10;10(7):3138-3150. doi: 10.7150/thno.43441. eCollection 2020. Theranostics. 2020. PMID: 32194859 Free PMC article.
-
Goals in Nutrition Science 2020-2025.Front Nutr. 2021 Feb 9;7:606378. doi: 10.3389/fnut.2020.606378. eCollection 2020. Front Nutr. 2021. PMID: 33665201 Free PMC article. Review.
-
Metabolic regulators of enigmatic inflammasomes in autoimmune diseases and crosstalk with innate immune receptors.Immunology. 2021 Aug;163(4):348-362. doi: 10.1111/imm.13326. Epub 2021 May 2. Immunology. 2021. PMID: 33682108 Free PMC article. Review.
-
NLRX1 Deficiency Alters the Gut Microbiome and Is Further Exacerbated by Adherence to a Gluten-Free Diet.Front Immunol. 2022 Apr 28;13:882521. doi: 10.3389/fimmu.2022.882521. eCollection 2022. Front Immunol. 2022. PMID: 35572547 Free PMC article.
References
-
- Coutermarsh-Ott S, Simmons A, Capria V, LeRoith T, Wilson JE, Heid B, Philipson CW, Qin Q, Hontecillas-Magarzo R, Bassaganya-Riera J, Ting JP, Dervisis N, Allen IC. NLRX1 suppresses tumorigenesis and attenuates histiocytic sarcoma through the negative regulation of NF-kappaB signaling. Oncotarget. 2016 - PMC - PubMed
-
- Moore CB, Bergstralh DT, Duncan JA, Lei Y, Morrison TE, Zimmermann AG, Accavitti-Loper MA, Madden VJ, Sun L, Ye Z, Lich JD, Heise MT, Chen Z, Ting JP. NLRX1 is a regulator of mitochondrial antiviral immunity. Nature. 2008;451(7178):573–577. - PubMed
-
- Allen IC, Moore CB, Schneider M, Lei Y, Davis BK, Scull MA, Gris D, Roney KE, Zimmermann AG, Bowzard JB, Ranjan P, Monroe KM, Pickles RJ, Sambhara S, Ting JP. NLRX1 protein attenuates inflammatory responses to infection by interfering with the RIG-I-MAVS and TRAF6-NF-kappaB signaling pathways. Immunity. 2011;34(6):854–865. - PMC - PubMed
-
- Guo H, Konig R, Deng M, Riess M, Mo J, Zhang L, Petrucelli A, Yoh SM, Barefoot B, Samo M, Sempowski GD, Zhang A, Colberg-Poley AM, Feng H, Lemon SM, Liu Y, Zhang Y, Wen H, Zhang Z, Damania B, Tsao LC, Wang Q, Su L, Duncan JA, Chanda SK, Ting JP. NLRX1 Sequesters STING to Negatively Regulate the Interferon Response, Thereby Facilitating the Replication of HIV-1 and DNA Viruses. Cell Host Microbe. 2016;19(4):515–528. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

