Cell fate decisions emerge as phages cooperate or compete inside their host
- PMID: 28165024
- PMCID: PMC5303824
- DOI: 10.1038/ncomms14341
Cell fate decisions emerge as phages cooperate or compete inside their host
Abstract
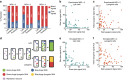
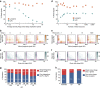
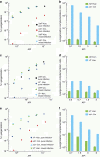
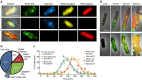
The system of the bacterium Escherichia coli and its virus, bacteriophage lambda, is paradigmatic for gene regulation in cell-fate development, yet insight about its mechanisms and complexities are limited due to insufficient resolution of study. Here we develop a 4-colour fluorescence reporter system at the single-virus level, combined with computational models to unravel both the interactions between phages and how individual phages determine cellular fates. We find that phages cooperate during lysogenization, compete among each other during lysis, and that confusion between the two pathways occasionally occurs. Additionally, we observe that phage DNAs have fluctuating cellular arrival times and vie for resources to replicate, enabling the interplay during different developmental paths, where each phage genome may make an individual decision. These varied strategies could separate the selection for replication-optimizing beneficial mutations during lysis from sequence diversification during lysogeny, allowing rapid adaptation of phage populations for various environments.
Conflict of interest statement
The authors declare no competing financial interests.
Figures


Similar articles
-
Fine-tuned spatiotemporal dynamics of DNA replication during phage lambda infection.J Virol. 2024 Nov 19;98(11):e0112824. doi: 10.1128/jvi.01128-24. Epub 2024 Oct 31. J Virol. 2024. PMID: 39480083 Free PMC article.
-
Interactions between Viral Regulatory Proteins Ensure an MOI-Independent Probability of Lysogeny during Infection by Bacteriophage P1.mBio. 2021 Oct 26;12(5):e0101321. doi: 10.1128/mBio.01013-21. Epub 2021 Sep 14. mBio. 2021. PMID: 34517752 Free PMC article.
-
Lysis-lysogeny coexistence: prophage integration during lytic development.Microbiologyopen. 2017 Feb;6(1):e00395. doi: 10.1002/mbo3.395. Epub 2016 Aug 17. Microbiologyopen. 2017. PMID: 27530202 Free PMC article.
-
Phage λ--new insights into regulatory circuits.Adv Virus Res. 2012;82:155-78. doi: 10.1016/B978-0-12-394621-8.00016-9. Adv Virus Res. 2012. PMID: 22420854 Review.
-
Single-Cell Studies of Phage λ: Hidden Treasures Under Occam's Rug.Annu Rev Virol. 2016 Sep 29;3(1):453-472. doi: 10.1146/annurev-virology-110615-042127. Epub 2016 Jul 22. Annu Rev Virol. 2016. PMID: 27482899 Review.
Cited by
-
Lytic/Lysogenic Transition as a Life-History Switch.Virus Evol. 2024 Apr 3;10(1):veae028. doi: 10.1093/ve/veae028. eCollection 2024. Virus Evol. 2024. PMID: 38756985 Free PMC article.
-
Cellular and Natural Viral Engineering in Cognition-Based Evolution.Commun Integr Biol. 2023 May 2;16(1):2196145. doi: 10.1080/19420889.2023.2196145. eCollection 2023. Commun Integr Biol. 2023. PMID: 37153718 Free PMC article. Review.
-
Temporal encoding of bacterial identity and traits in growth dynamics.Proc Natl Acad Sci U S A. 2020 Aug 18;117(33):20202-20210. doi: 10.1073/pnas.2008807117. Epub 2020 Aug 3. Proc Natl Acad Sci U S A. 2020. PMID: 32747578 Free PMC article.
-
A Protocol to Engineer Bacteriophages for Live-Cell Imaging of Bacterial Prophage Induction Inside Mammalian Cells.STAR Protoc. 2020 Aug 7;1(2):100084. doi: 10.1016/j.xpro.2020.100084. eCollection 2020 Sep 18. STAR Protoc. 2020. PMID: 33111117 Free PMC article.
-
Infection by bacteriophage lambda: an evolving paradigm for cellular individuality.Curr Opin Microbiol. 2018 Jun;43:9-13. doi: 10.1016/j.mib.2017.09.014. Epub 2017 Nov 3. Curr Opin Microbiol. 2018. PMID: 29107897 Free PMC article. Review.
References
-
- Mcfarland D. J. Decision-making in animals. Nature 269, 15–21 (1977).
-
- Cotman C. Apoptosis decision cascades and degeneration in Alzheimers disease. Neurobiol. Aging 19, 29–32 (1998). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

