Chromatographic Separation of Vitamin E Enantiomers
- PMID: 28165404
- PMCID: PMC6155869
- DOI: 10.3390/molecules22020233
Chromatographic Separation of Vitamin E Enantiomers
Abstract
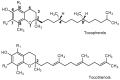
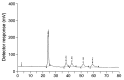
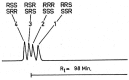
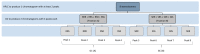
Vitamin E is recognized as an essential vitamin since its discovery in 1922. Most vegetable oils contain a mixture of tocopherols and tocotrienols in the vitamin E composition. Structurally, tocopherols and tocotrienols share a similar chromanol ring and a side chain at the C-2 position. Owing to the three chiral centers in tocopherols, they can appear as eight different stereoisomers. Plant sources of tocopherol are naturally occurring in the form of RRR while synthetic tocopherols are usually in the form of all-racemic mixture. Similarly, with only one chiral center, natural tocotrienols occur as the R-isoform. In this review, we aim to discuss a few chromatographic methods that had been used to separate the stereoisomers of tocopherols and tocotrienols. These methods include high performance liquid chromatography, gas chromatography and combination of both. The review will focus on method development including selection of chiral columns, detection method and choice of elution solvent in the context of separation efficiency, resolution and chiral purity. The applications for separation of enantiomers in vitamin E will also be discussed especially in terms of the distinctive biological potency among the stereoisoforms.
Keywords: Vitamin E; chromatography; enantiomers; tocopherols; tocotrienols.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies) Scientific Opinion on Dietary Reference Values for vitamin E as α-tocopherol. EFSA J. 2015;13:4149.
-
- Goh S.H., Choo Y.M., Ong S.H. Minor constituents of palm oil. J. Am. Oil Chem. Soc. 1985;62:237–240. doi: 10.1007/BF02541384. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

