Depletion of AADC activity in caudate nucleus and putamen of Parkinson's disease patients; implications for ongoing AAV2-AADC gene therapy trial
- PMID: 28166239
- PMCID: PMC5293261
- DOI: 10.1371/journal.pone.0169965
Depletion of AADC activity in caudate nucleus and putamen of Parkinson's disease patients; implications for ongoing AAV2-AADC gene therapy trial
Abstract
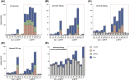
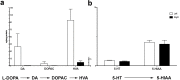
In Parkinson's disease (PD), aromatic L-amino acid decarboxylase (AADC) is the rate-limiting enzyme in the conversion of L-DOPA (Sinemet) to dopamine (DA). Previous studies in PD animal models demonstrated that lesion of dopaminergic neurons is associated with profound loss of AADC activity in the striatum, blocking efficient conversion of L-DOPA to DA. Relatively few studies have directly analyzed AADC in PD brains. Thus, the aim of this study was to gain a better understanding of regional changes in AADC activity, DA, serotonin and their monoamine metabolites in the striatum of PD patients and experimentally lesioned animals (rat and MPTP-treated nonhuman primate, NHP). Striatal AADC activity was determined post mortem in neuropathologically confirmed PD subjects, animal models and controls. A regional analysis was performed for striatal AADC activity and monoamine levels in NHP tissue. Interestingly, analysis of putaminal AADC activity revealed that control human striatum contained much less AADC activity than rat and NHP striata. Moreover, a dramatic loss of AADC activity in PD striatum compared to controls was detected. In MPTP-treated NHP, caudate nucleus was almost as greatly affected as putamen, although mean DA turnover was higher in caudate nucleus. Similarly, DA and DA metabolites were dramatically reduced in different regions of PD brains, including caudate nucleus, whereas serotonin was relatively spared. After L-DOPA administration in MPTP-treated NHP, very poor conversion to DA was detected, suggesting that AADC in NHP nigrostriatal fibers is mainly responsible for L-DOPA to DA conversion. These data support further the rationale behind viral gene therapy with AAV2-hAADC to restore AADC levels in putamen and suggest further the advisability of expanding vector delivery to include coverage of anterior putamen and the caudate nucleus.
Conflict of interest statement
KSB is a Scientific Founder of Voyager Therapeutics, company that is involved in developing AAV2-AADC gene therapy for Parkinson's Disease. All authors declare that they have no competing interests relating to patents, products in development, or marketed products.
Figures


References
-
- Aminoff MJ. Parkinson's disease. Neurol Clin. 2001;19(1):119–28, vi. - PubMed
-
- Goldstein DJ. Parkinson's disease. Arch Neurol. 1988;45(7):715–6. - PubMed
-
- Poewe W, Mahlknecht P. The clinical progression of Parkinson's disease. Parkinsonism Relat Disord. 2009;15 Suppl 4:S28–32. - PubMed
-
- Skirboll S, Wang J, Mefford I, Hsiao J, Bankiewicz KS. In vivo changes of catecholamines in hemiparkinsonian monkeys measured by microdialysis. Exp Neurol. 1990;110(2):187–93. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

